The change in the internal energy of an ideal gas does not depend on?
1.
Number of moles
2.
Change in temperature
3.
Specific heat at constant pressure \(C_p\) of the gas
4.
Specific heat at constant volume \(C_v\) of the gas
The translational kinetic energy of \(n\) moles of a diatomic gas at absolute temperature \(T\) is given by:
1. \(\frac{5}{2}nRT\)
2. \(\frac{3}{2}nRT\)
3. \(5nRT\)
4. \(\frac{7}{2}nRT\)
Which of the following graphs, shows the variation of the mean kinetic energy \(E\) of an ideal gas molecule with temperature
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | \(3:1\) | 2. | \(9:1\) |
| 3. | \(1:1\) | 4. | \(1:4\) |
When a large bubble rises from the bottom of a lake to the surface, its radius doubles. The atmospheric pressure is equal to that of a column of water of height H. The depth of the lake is:
1. H
2. 2H
3. 7H
4. 8H
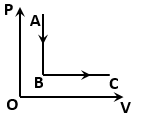
The figure shows a process for a gas in which pressure (P) and volume (V) of the gas change. If and are the molar heat capacities of the gas during the processes AB and BC respectively, then:
1.
2.
3.
4.
The pressure in a diatomic gas increases from to , when its volume is increased from . The increase in internal energy will be:
1.
2.
3.
4.
If at a pressure of \(10^6\) dyne/cm2, one gram of nitrogen occupies \(2\times10^4\) c.c. volume, then the average energy of a nitrogen molecule in erg is:
| 1. | \(14\times10^{-13}\) | 2. | \(10\times10^{-12}\) |
| 3. | \(10^{6}\) | 4. | \(2\times10^{6}\) |
Without change in temperature, a gas is forced in a smaller volume. Its pressure increases because its molecules:
| 1. | strike the unit area of the container wall more often. |
| 2. | strike the unit area of the container wall at a higher speed. |
| 3. | strike the unit area of the container wall with greater force. |
| 4. | have more energy. |
If the mean free path of atoms is doubled, then the pressure of the gas will become:
1. \(\frac{P}{4}\)
2. \(\frac{P}{2}\)
3. \(\frac{P}{8}\)
4. \(P\)