| 1. |  |
2. |  |
| 3. | 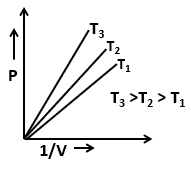 |
4. | - |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology| 1. | STP | 2. | 0°C, 2 atm |
| 3. | 0°C, 4 atm | 4. | 273°C, 4 atm |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology(Given - molar volume at STP is 22.4 L)
1. 6.72
2. 2.24
3. 22.4
4. 67.2
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyChoose the correct option for graphical representation of Boyle's law, which shows a graph of pressure vs. volume of a gas at different temperatures:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyA mixture of and Ar gases in a cylinder contains 7 g of and 8 g of Ar. If the total pressure of the mixture of the gases in the cylinder is 27 bar, the partial pressure of is:
[Use atomic masses (in g ): N=14, Ar=40]
1. 12 bar
2. 15 bar
3. 18 bar
4. 9 bar
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe minimum pressure required to compress 600 dm3 of a gas at 1 bar to 150 dm3 at 40°C is
1. 4.0 bar
2. 0.2 bar
3. 1.0 bar
4. 2.5 bar
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology