The electrode potential for Mg electrode varies according to the equation
\(E_{Mg^{2+}/Mg}\ = \ E_{Mg^{2+}/Mg}^{o} \ - \ \frac{0.059}{2}log\frac{1}{[Mg^{2+}]}\)
The graph of EMg2+ / Mg vs log [Mg2+] among the following is:
| 1. | 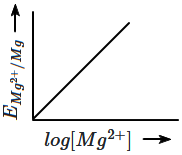 |
2. |  |
| 3. |  |
4. |  |
Subtopic: Nernst Equation |
60%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
= 1.1 V for Daniel cell. Which of the following expressions are correct descriptions of the state of equilibrium in this cell?
(a) 1.1 = KC
(b)
(c)
(d) log KC = 1.1
The correct choice among the given is -
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
Subtopic: Nernst Equation |
85%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
For the given cell, Mg | Mg2+ || Cu2+ | Cu
a. Mg acts as cathode
b. Cu acts as cathode
c. The cell reaction is Mg+ Cu2+ —-> Mg2+ + Cu
d. Cu is the oxidising agent
The correct choice among the given is -
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
Subtopic: Electrolytic & Electrochemical Cell | Nernst Equation |
74%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


