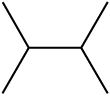
Match the item in column I with the column II
Column I
Column II
a.
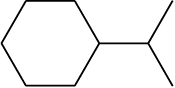
i.

b.

ii.

c.
iii.

d.
iv.

1. a-ii; b-iv; c-i; d-iii
2. a-iii; b-iv; c-ii; d-i
3. a-iv; b-iii; c-i; d-ii
4. a-i; b-iv; c-ii; d-iii







2. a-iii; b-iv; c-ii; d-i
3. a-iv; b-iii; c-i; d-ii
4. a-i; b-iv; c-ii; d-iii
Incorrect match amongst the following is/are-
| (a) |  |
3,4,7-trimethyloctane |
| (b) |  |
5-ethyl-3-methylheptane |
| (c) |  |
2,3,4-trimethylheptane |
1. a, c
2. a, b
3. b, c
4. Only b
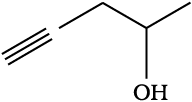
The correct IUPAC name of the given compound is-

1. 6-Ethylhexan-3-ol
2. 2-Ethylhexan-4-ol
3. 3-Methyloctan-6-ol
4. 6-Methyloctan-3-ol
The IUPAC name of the given compound is-
1. Hexane-3,5-dione
2. Hexane-2,4-dione
3. Hex-2,4-dione
4. Hex-3,5-dione
The IUPAC name of the given compound is-
1. Hexa-1,3-dien-5-yne
2. Hexa-3,5-dien-1-yne
3. Hexa-1,3-en-5-yne
4. Hexane-1,3-dien-5-yne
Match the structures given in column I with the corresponding IUPAC names given in column II and choose the correct option.
| Column I | Column II |
A.  |
i. Pent-4-en-2-ol |
B.  |
ii. Cyclohex-2-en-1-ol |
C.  |
iii. 6-Hydroxy-heptanal |
D.  |
iv. 2-Chlorohexane |
E.  |
v. 3- Nitrocyclohexene |
1. A=iii; B= iv; C=v; D=ii; E=i
2. A=iii; B= iv; C=ii; D=v; E=i
3. A=iv; B= i; C=v; D=iii; E=ii
4. A=iv; B= i; C=ii; D=v; E=iii
Match the items in column I with the items in column II.
| Column I | Column II |
| a. o-Ethylanisole | i.  |
| b. p-Nitroaniline | ii.  |
| c. 2,3 - Dibromo -1 - phenylpentane | iii.  |
| d. 4-Ethyl-1-fluoro-2-nitrobenzene | iv.  |
The correct option is-
1. a=ii; b=iv; c=i; d=iii
2. a=ii; b=i; c=iv; d=iii
3. a=iv; b=i; c=ii; d=iii
4. a=ii; b=iii; c=iv; d=i
The incorrect curved-arrow notation that shows the formation of reactive intermediates in the following compound is-
| 1. |  |
| 2. | 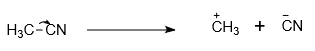 |
| 3. | 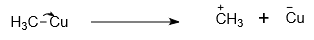 |
| 4. | All of the above |
| Column I (Molecules) |
COLUMN II (Nature of molecule) |
| a. (CH3)3N: | i. Electrophile |
| b. \(H_3C-\overset{+}{C}=O\) | ii. Nucleophile |
| c. \(\overset{+}{N}O_2\) | |
| d. H2N:- |
| a | b | c | d | |
| 1. | ii | ii | ii | i |
| 2. | ii | ii | ii | ii |
| 3. | ii | i | i | ii |
| 4. | ii | i | ii | ii |
Consider the following molecule.
(a) H3C-H, H3C-Br
(b) H3C-NH2, H3C-OH
(c) H3C-OH, H3C-SH
The bond which is more polar in the given pairs of molecules is:
| 1. | C-H bond is more polar than C-Br bond; C-O is more polar than C-N and C-S bond |
| 2. | C-Br bond is more polar than C-H bond; C-O is more polar than C-N and C-S bond |
| 3. | C-Br bond is more polar than C-H bond; C-N is more polar than C-O and C-O bond is more polar than C-S |
| 4. | None of the above |



