The correct reactivity order towards hydrolysis is:
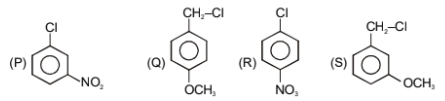
1.
Q>R>S>P
2.
Q>P>R>S
3.
S>R>Q>P
4.
Q>S>R>P

The effect that makes 2,3–dimethyl-2-butene more stable than 2-butene is-
1. Resonance
2. Hyperconjugation
3. Steric effect
4. Inductive effect
Which types of isomerism can be exhibited by compounds with the molecular formula C4H11N?
1. Position isomerism
2. Metamerism
3. Functional isomerism
4. All of the above

The IUPAC name of the above mentioned compound is -
1. Cyclohexylidenemethanone
2. Cyclohexylidemethanone
3. Cyclohexylidenylmethanone
4. Cyclohexdenemethanone
A tertiary butyl carbocation is more stable than a secondary butyl carbocation because-
1. + R effect of groups
2. R effect of CH3 groups
3. Hyperconjugation
4. l effect of CH3 groups
1. 2–Bromo-3–methylbutanoic acid
2. 2-Methyl-3-bromobutanoic acid
3. 3-Bromo-2-methylbutanoic acid
4. 3-Bromo-2,3-dimethylpropanoic acid.
The pair of structures that does not represent isomers is:
| 1. | 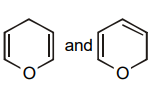 |
2. | 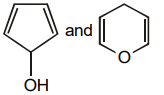 |
| 3. | 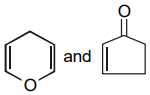 |
4. | 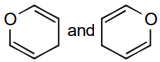 |
Which carbocation is the most stable among the options provided?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The aromatic compound among the following is:-
| 1. | 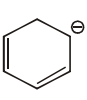 |
2. |  |
| 3. | 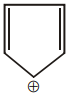 |
4. | 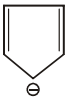 |
The pair among the following that does not contain position isomers is -
| 1. | 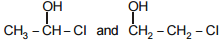 |
| 2. | 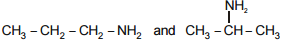 |
| 3. |  |
| 4. |  |



