The correct statement regarding the comparison of staggered and eclipsed conformations of ethane is:
| 1. | The eclipsed conformation of ethane is more stable than staggered conformation because eclipsed conformation has no torsional strain. |
| 2. | The eclipsed conformation of ethane is more stable than staggered conformation even though the eclipsed conformation has a torsional strain. |
| 3. | The staggered conformation of ethane is more stable than eclipsed conformation because staggered conformation has no torsional strain. |
| 4. | The staggered conformation of ethane is less stable than eclipsed conformation because staggered conformation has a torsional strain. |
An unsaturated hydrocarbon 'A' reacts with two molecules of H2 and upon reductive ozonolysis A gives butane-1,4-dial, ethanal, and propanone.
The IUPAC name of A is:
1. 2-Methylocta-2,6-diene
2. 2-Methylocta-1,5-diene
3. 3-Methylocta-2,6-diene
4. 2-Methylocta-1,6-diene
The most suitable reagent for the following conversion is-

1. Hg2+/ H+, H2O
2. Na/liquid NH3
3. H2, Pd/C, quinoline
4. Zn/HCl
Product Z in the above-mentioned reaction is:
1.
2.
3.
4.
The correct IUPAC name of the given compound is:
1. 3-Ethyl-4-ethenylheptane
2. 3-Ethyl-4-propylhex-5-ene
3. 3-(1-Ethyl propyl) hex-1-ene
4. 4-Ethyl-3-propylhex-1-ene
The incorrect IUPAC name among the following is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
The isobutyl group among the following is:
| 1. | |
| 2. | CH3–CH2–CH2–CH2– |
| 3. | 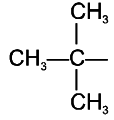 |
| 4. |
If Compound A (C₄H₈) is treated with H₂O/H₂SO₄ and forms an optically inactive C₄H₁₀O, what is the structure of A?
| 1. | CH3CH2CH=CH2 | 2. | CH3CH=CHCH3 |
| 3. | (CH3)2C=CH2 | 4. |
Which of the following statement(s) is/are correct about the elimination reaction of 2-Bromopentane to form pent-2-ene:
(a) -Elimination reaction
(b) Follows Zaitsev rule
(c) Dehydrohalogenation reaction
(d) Dehydration reaction
| 1. | (a), (c), (d) | 2. | (b), (c), (d) |
| 3. | (a), (b), (d) | 4. | (a), (b), (c) |
The dihedral angle of the least stable conformer of ethane is:
| 1. | 60° | 2. | 0° |
| 3. | 120° | 4. | 180° |




