Match List I with List II.
Choose the correct answer from the options given below :
1. \(\text { A-III, B-I, C-II, D-IV }\)
2. \(\text { A-IV, B-I, C-II, D-III }\)
3. \(\text { A-I, B-IV, C-II, D-III }\)
4. \(\text { A-IV,B-I, C-III, D-II }\)
| List I (Reaction) |
List II (Reagents/ Condition) |
A.  |
I.  |
B. |
II.\(\mathrm{CrO}_3\) |
C.  |
III.\(\begin{aligned} & \mathrm{KMnO}_4 \\ & \mathrm{KOH}, \Delta \end{aligned}\) |
D.  |
IV. \(\mathrm{(i) \mathrm{O}_3\\ (ii) \mathrm{Zn}-\mathrm{H}_2 \mathrm{O}}\) |
Choose the correct answer from the options given below :
1. \(\text { A-III, B-I, C-II, D-IV }\)
2. \(\text { A-IV, B-I, C-II, D-III }\)
3. \(\text { A-I, B-IV, C-II, D-III }\)
4. \(\text { A-IV,B-I, C-III, D-II }\)
Subtopic: Aldehydes & Ketones: Preparation & Properties | Carboxylic Acids: Preparation & Properties |
69%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Fehling solution 'A' is:
| 1. | Alkaline copper sulphate |
| 2. | Alkaline solution of sodium potassium tartrate (Rochelle's salt) |
| 3. | Aqueous sodium citrate |
| 4. | Aqueous copper sulphate |
Subtopic: Aldehydes & Ketones: Preparation & Properties |
52%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Match List-I with List-II
Choose the correct answer from the options given below:
1. A-II, B-III, C-I, D-IV
2. A-IV, B-III, C-I, D-II
3. A-I, B-III, C-IV, D-II
4. A-II, B-I, C-III, D-IV
| List-I (Reagent) |
List-II (Name of the reaction) |
||
| A. | \(\mathrm{{H}_2, {Nd}-{Ba}S {O}_4}\) | I. | Gattermann-koch reaction |
| B. | \(\mathrm{{(i) \; {CrO}_2 {Cl}_2, {CS}_2\\ (ii) {H}_2 {C}}}\) | II. | Reimer-Tiemann reaction |
| C. | \(\mathrm{{CO, HCl, Anhyd.~\\ {AlCl}_3 / {CuCl}}}\) | III. | Etard reaction |
| D. | \(\mathrm{{CHCl}_3, {NaOH}}\) | IV. | Rosenmund reduction |
1. A-II, B-III, C-I, D-IV
2. A-IV, B-III, C-I, D-II
3. A-I, B-III, C-IV, D-II
4. A-II, B-I, C-III, D-IV
Subtopic: Aldehydes & Ketones: Preparation & Properties |
89%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Identify [X] and [Y] in given reaction:


| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Subtopic: Aldehydes & Ketones: Preparation & Properties |
67%
From NCERT
NEET - 2024
Please attempt this question first.
Hints
Please attempt this question first.
Given is a reaction for your reference:
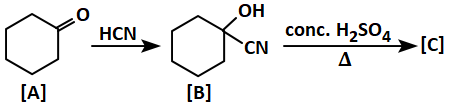
The final product [C] is:

The final product [C] is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Aldehydes & Ketones: Preparation & Properties | Carboxylic Acids: Preparation & Properties |
61%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Identify the final product [D] obtained in the following sequence of reactions:




| 1. | \(\mathrm{HC\equiv C^-~Na^+}\) | 2. |  |
| 3. |  |
4. | \(\mathrm{C_4H_{10}}\) |
Subtopic: Aldehydes & Ketones: Preparation & Properties |
73%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
Given below are two statements:
In the light of the above statements, choose the most appropriate answer from the options given below:
| Statement I | The boiling points of aldehydes and ketones are higher than hydrocarbons of comparable molecular masses because of weak molecular association in aldehydes and ketones due to dipole-dipole interactions. |
| Statement II | The boiling points of aldehydes and ketones are lower than the alcohols of similar molecular masses due to the absence of H-bonding. |
In the light of the above statements, choose the most appropriate answer from the options given below:
| 1. | Statement I is incorrect but Statement II is correct. |
| 2. | Both Statement I and Statement II are correct. |
| 3. | Both Statement I and Statement II are incorrect. |
| 4. | Statement I is correct but Statement II is incorrect. |
Subtopic: Aldehydes & Ketones: Preparation & Properties |
63%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The correct IUPAC name of the following compound is:

| 1. | 2-Ethylhex-3-en-4-one | 2. | 4-Methylhex-3-en-2-one |
| 3. | 4-Ethylpent-3-en-2-one | 4. | 3-Methylhex -3-en-4-one |
Subtopic: Aldehydes & Ketones: Preparation & Properties |
86%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
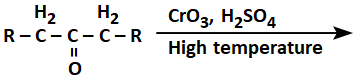
The product(s) formed from the following reaction is/are:
| 1. | RCOOH only |
| 2. | RCH2COOH only |
| 3. | 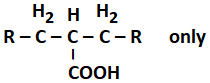 |
| 4. | RCOOH and RCH2COOH |
Subtopic: Aldehydes & Ketones: Preparation & Properties |
69%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The product formed from the following reaction sequence is:


| 1. |  |
2. |  |
| 3. |  |
4. |  |
Subtopic: Aldehydes & Ketones: Preparation & Properties | Carboxylic Acids: Preparation & Properties | Name Reaction |
61%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch