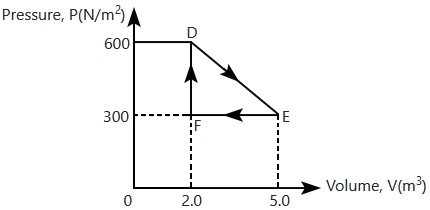
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in the figure. Its volume is then reduced to the original value from \(\mathrm{E}\) to \(\mathrm{F}\) by an isobaric process. The total work done by the gas from \(\mathrm{D}\) to \(\mathrm{E}\) to \(\mathrm{F}\) is:
1. \(600\) J
2. \(300\) J
3. \(450\) J
4. \(500\) J
Subtopic: Work Done by a Gas |
84%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch