The IUPAC name of the above compound is -
1. 4 – Ethoxypropane
2. 3 – Ethoxypropane
3. 1 – Ethoxypropane
4. 2 – Ethoxypropane
The IUPAC name of the following compound is -
1. 1-Ethoxy-3-methylpentane
2. 3-Ethoxy-1-methylpentane
3. 2-Ethoxy-3-methylpentane
4. 3-Ethoxy-2-methylpentane
The structure and name of intermediate compound is -
The IUPAC name of
is -
1. Cyclopent-1-en-3-ol
2. Cyclopent-3-en-1-ol
3. Cyclopent-2-en-1-ol
4. Cyclopent-1-en-3-ol
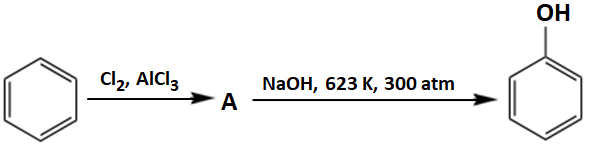
"A" in the reaction below is:
| 1. | Chlorobenzene | 2. | 1,2-Dichlorobenzene |
| 3. | 1,3-dichlorobenzene | 4. | None of these |
The IUPAC name of above mentioned compound is -
1. 1-Chloro-3-ethylbutan-3-ol
2. 1-Chloro-3-ethylbutan-1-ol
3. 3-Chloro-3-ethylbutan-1-ol
4. 3-(chloromethyl) pentan-1-ol
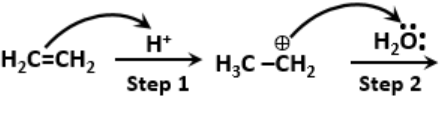
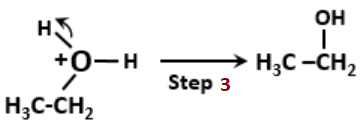
The proposed mechanism for hydration of ethene to yield ethanol is as follows:
The wrong step in the above mechanism is-
1. Step 1
2. Step 2
3. Step 3
4. Both steps 2 and 3
The reaction of 3-methylbutan-2-ol with HBr gives -
| 1. | 2-Bromo-3-methyl butane | 2. | 2-Bromo-2-methyl butane |
| 3. | 3-Bromo-2-methyl butane | 4. | 3-Bromo-3-methyl butane |
The reaction of hydrogen iodide with methoxybenzene gives -
1. Phenol
2. Iodomethane
3. Phenol and Iodomethane
4. None of the these
The reaction of hydrogen iodide with benzyl ethyl ether gives -
1. Ethanol
2. Benzyl iodide
3. Benzyl iodide and Ethanol
4. None of the above