Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly
A gas \((\gamma = 1.5)\) undergoes a process in which its volume is doubled, but the speed of sound in the gas remains unchanged. Then,
| 1. | the pressure is halved |
| 2. | the pressure decreases by a factor of \(2\sqrt 2\) |
| 3. | the temperature is halved |
| 4. | the temperature decreases by a factor of \(2 \sqrt 2\) |
Subtopic: Types of Processes |
51%
Level 3: 35%-60%
Hints
The pressure of an ideal gas \(\left(\gamma=\dfrac32\right)\) is increased by \(1\%\) in an adiabatic process. The temperature of the gas:
| 1. | increases by \(1.5\%\) |
| 2. | decreases by \(1.5\%\) |
| 3. | increases by \(\frac13\%\) |
| 4. | increases by \(\frac23\%\) |
Subtopic: Types of Processes |
61%
Level 2: 60%+
Hints
Unlock IMPORTANT QUESTION
This question was bookmarked by 5 NEET 2025 toppers during their NEETprep journey. Get Target Batch to see this question.
✨ Perfect for quick revision & accuracy boost
Buy Target Batch
Access all premium questions instantly
An ideal monoatomic gas at a temperature of \(300\) K and a pressure of \(10\) atm is suddenly allowed to expand into vacuum so that its volume is doubled. No exchange of heat is allowed to take place between the gas and its surroundings during the process. After equilibrium is reached, the final temperature is:
| 1. | \(300\) K | 2. | \(\dfrac{300}{2^{5/3}}\) K |
| 3. | \(\dfrac{300}{2^{2/3}}\) K | 4. | \(600\) K |
Subtopic: Types of Processes |
Level 3: 35%-60%
Hints
One mole of an ideal gas goes from an initial state \(A\) to the final state \(B\) with two processes. It first undergoes isothermal expansion from volume \(V\) to \(3V\) and then its volume is reduced from \(3V\) to \(V\) at constant pressure. The correct \((P-V)\) diagram representing the two processes is:
| 1. |  |
2. | 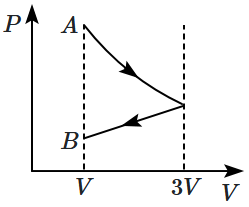 |
| 3. |  |
4. |  |
Subtopic: Types of Processes |
82%
Level 1: 80%+
AIPMT - 2012
Hints
Links
Which of the following relations does not give the equation of an adiabatic process, where terms have their usual meaning?
1. \({P}^{1-\gamma}{T}^{\gamma}= \text{constant}\)
2. \({PV}^{\gamma}=\text{constant}\)
3. \({TV}^{\gamma-1}= \text{constant}\)
4. \({P}^{\gamma} {T}^{1-\gamma}=\text{constant}\)
1. \({P}^{1-\gamma}{T}^{\gamma}= \text{constant}\)
2. \({PV}^{\gamma}=\text{constant}\)
3. \({TV}^{\gamma-1}= \text{constant}\)
4. \({P}^{\gamma} {T}^{1-\gamma}=\text{constant}\)
Subtopic: Types of Processes |
76%
Level 2: 60%+
NEET - 2013
Hints
The ratio \(C_P/C_V=1.5\) for a certain ideal gas. The gas is taken at an initial pressure of \(2\) kPa and compressed suddenly to \(\dfrac14\) of its initial volume. The final pressure is:
1. \(\dfrac12\) kPa
2. \(4\) kPa
3. \(8\) kPa
4. \(16\) kPa
Subtopic: Types of Processes |
71%
Level 2: 60%+
Hints
If heat is supplied to an ideal gas in an isothermal process,
| 1. | the internal energy of the gas will increase. |
| 2. | the gas will do positive work. |
| 3. | the gas will do negative work. |
| 4. | the said process is not possible. |
Subtopic: Types of Processes |
76%
Level 2: 60%+
Hints
In thermodynamic processes which of the following statements is not true?
| 1. | In an adiabatic process, the system is insulated from the surroundings. |
| 2. | In an isochoric process, pressure remains constant. |
| 3. | In an isothermal process, the temperature remains constant. |
| 4. | In an adiabatic process \(PV^\gamma=\mathrm{constant}.\) |
Subtopic: Types of Processes |
92%
Level 1: 80%+
AIPMT - 2009
Hints




