Combustion of glucose takes place according to the equation,
,
Energy required for the production of 1.6 g of glucose is -
(Molecular mass of glucose = 180 g)
1. 0.064 kcal
2. 0.64 kcal
3. 6.4 kcal
4. 64 kcal
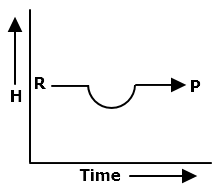
Which plot represents an exothermic reaction?
1.
2.
3.
4.
The enthalpy and entropy change for the reaction :
Br2 (l) + Cl2 (g) 2BrCl (g)
are 30 kJ mol-1 and 105 J K-1 mol-1 respectively.
The temperature at which the reaction will be in equilibrium is :
| 1. | 285.7 K | 2. | 273.4 K |
| 3. | 450.9 K | 4. | 300.1 K |
For the reaction, , at 298K is 164 kJ mol-1. The of the reaction is-
1. \(166.5 \mathrm{~kJ} \mathrm{~mol}^{-1} \)
2. \(141.5 \mathrm{~kJ} \mathrm{~mol}^{-1} \)
3. \(104.0 \mathrm{~kJ} \mathrm{~mol}^{-1} \)
4. \(-169 \mathrm{~kJ} \mathrm{~mol}^{-1}\)
The bond energies of , C-H, H-H, and C=C are 198, 98, 103 and145 kcal respectively.
The enthalpy change of the reaction would be-
1. 48 kcal
2. 96 kcal
3. -40 kcal
4. -152 kcal
Which among the following state functions is an extensive property of the system?
| 1. | Temperature | 2. | Volume |
| 3. | Refractive index | 4. | Viscosity |
Which, among the following, is not a state function?
1. Internal energy
2. Free energy
3. Work
4. Enthalpy
Which of the following is correct for isothermal expansion of an ideal gas:
1. Wrev = Wirr
2. Wrev + Wirr = 0
3. Wrev > Wirr
4. qrev = qirr
The correct statement for a reversible process in a state of equilibrium is:
1. G = – 2.30RT log K
2. G = 2.30RT log K
3. Go = – 2.30RT log K
4. Go = 2.30RT log K
From the following bond energies:
H—H bond energy: 431.37 kJ mol-1
C=C bond energy: 606.10 kJ mol-1
C—C bond energy: 336.49 kJ mol-1
C—H bond energy: 410.50 kJ mol-1
Enthalpy for the reaction,
will be:
| 1. | 1523.6 kJ mol-1 | 2. | -243.6 kJ mol-1 |
| 3. | -120.0 kJ mol-1 | 4. | 553.0 kJ mol-1 |