Which amongst the following options is correct graphical representation of Boyle's Law?
| 1. |  |
2. |  |
| 3. | 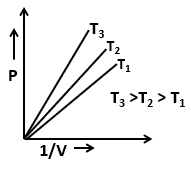 |
4. | - |
Subtopic: Gas Laws |
54%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
The correct option in which the density of argon (Atomic mass =40) is highest:
| 1. | STP | 2. | 0°C, 2 atm |
| 3. | 0°C, 4 atm | 4. | 273°C, 4 atm |
Subtopic: Gas Laws |
58%
From NCERT
NEET - 2023
Please attempt this question first.
Hints
Please attempt this question first.
A vessel contains 3.2 g of dioxygen gas at STP (273.15 K and 1 atm pressure). The gas is now transferred to another vessel at a constant temperature, where pressure becomes one-third of the original pressure. The volume of the new vessel in L is:
(Given - molar volume at STP is 22.4 L)
1. 6.72
2. 2.24
3. 22.4
4. 67.2
(Given - molar volume at STP is 22.4 L)
1. 6.72
2. 2.24
3. 22.4
4. 67.2
Subtopic: Gas Laws |
50%
From NCERT
NEET - 2022
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Choose the correct option for graphical representation of Boyle's law, which shows a graph of pressure vs. volume of a gas at different temperatures:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Subtopic: Gas Laws |
75%
From NCERT
NEET - 2021
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
A mixture of and Ar gases in a cylinder contains 7 g of and 8 g of Ar. If the total pressure of the mixture of the gases in the cylinder is 27 bar, the partial pressure of is:
[Use atomic masses (in g ): N=14, Ar=40]
1. 12 bar
2. 15 bar
3. 18 bar
4. 9 bar
Subtopic: Gas Laws |
72%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The minimum pressure required to compress 600 dm3 of a gas at 1 bar to 150 dm3 at 40°C is
1. 4.0 bar
2. 0.2 bar
3. 1.0 bar
4. 2.5 bar
Subtopic: Gas Laws |
76%
From NCERT
NEET - 2020
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


