A. Tollen's reagent
B. Schiff's reagent
C. \(\text{HCN}\)
D. \(\text{NH}_2\text{OH}\)
E. \(\text{NaHSO}_3\)
Choose the correct options from the given below:-
| 1. | \(\text{A}\) and \(\text{D}\) | 2. | \(\text{B}\) and \(\text{E}\) |
| 3. | \(\text{E}\) and \(\text{D}\) | 4. | \(\text{B}\) and \(\text{C}\) |
Which one is not a D-sugar:
| 1. |  |
2. | 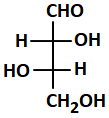 |
| 3. |  |
4. |  |
Which of the following statement is not true about glucose?
1. It is an aldohexose.
2. It contains five hydroxyl groups.
3. It is a reducing sugar.
4. It is an aldopentose.
2. Glucose
3. Sucrose
4. Maltose
D(+) glucose yields an oxime with hydroxyl amine. The structure of the oxime would be:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Sucrose can be formed by:
1. α–D–galactopyranose and α–D–glucopyranose
2. α–D–glucopyranose and β–D–fructofuranose
3. β–D–galactopyranose and α–D–fructofuranose
4. α–D–galactopyranose and β–D–fructopyranose
Fructose reduces Tollen's reagent due to:
| 1. | Primary alcoholic group |
| 2. | Secondary alcoholic group |
| 3. | Enolisation of fructose followed by conversion to aldehyde by base |
| 4. | Asymmetric carbon |
-D-glucose and β-D-glucose are:
1. Epimers
2. Anomers
3. Functional isomers
4. Chain isomers
Sucrose on hydrolysis gives:
1. L(+) Glucose + D(+) Fructose
2. L(–) Glucose + L(–) Fructose
3. D(+) Glucose + D(–) Fructose
4. D(+) Glucose + L(–) Fructose


