In this reaction,
\(CH_{3}CHO \ + \ HCN \ \xrightarrow[]{} \ CH_{3}CH(OH)CN \ \xrightarrow[]{\textbf{H.OH}} \\ CH_{3}CH(OH)COOH\)
an asymmetric compound is generated. The acid obtained would be:
1. 50% D + 50% L-isomer
2. 20% D + 80% L-isomer
3. D-isomer
4. L-isomer
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The Grignard reagent is added to methanal and hydrolyzed to produce:
| 1. | Primary alcohol | 2. | Tertiary alcohol |
| 3. | Methane | 4. | Methanoic acid |
The major product formed when cyclohexanecarbaldehyde reacts with PhMgBr and H3O+ is:
| 1. |  |
2. |  |
| 3. |  |
4. | None of these |
HCN not produce a chiral compound with:
| 1. | 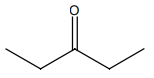 |
2. | 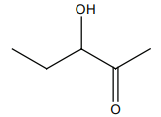 |
| 3. | 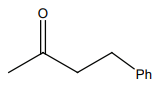 |
4. | 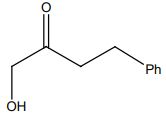 |
An alkene “A” on reaction with O3 and Zn - H2O gives propanone and ethanal in an equimolar ratio. The addition of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is:
| 1. | 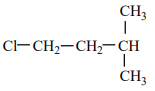 |
2. | 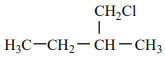 |
| 3. | 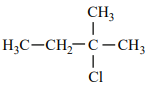 |
4. | 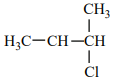 |
Which of the following compounds produces a pink colour when combined with Schiff's reagent?
1. CH3CHO
2. CH3CH2COOH
3. C3H5COOH
4. C6H5COC6H5
What is present in acetone's enolic form?
1. 9 -bonds, 1-bond and 2 lone pairs
2. 8 -bonds, 2-bonds and 2 lone pairs
3. 10 -bonds, 1-bond and 1 lone pair
4. 9 -bonds, 2-bonds and 1 lone pair
A compound A, when reacted with PCl5 and then with ammonia, gave B. B, when treated with bromine and caustic potash, produced C. C on treatment with NaNO2 and HCl at C and on boiling produced ortho-cresol. Compound A is:
| 1. | o-Toluic acid | 2. | o-Chlorotoluene |
| 3. | o-Bromotoluene | 4. | m-Toluic acid |
Select the correct option based on statements below:
| Assertion (A): | Compounds containing —CHO group are easily oxidised to corresponding carboxylic acids. |
| Reason (R): | Carboxylic acids can be reduced to alcohol by treatment with LiAlH4. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |


