Match the polymers given in Column-I with their repeating units given in Column-II. Choose the correct option from the codes given below :
Column-I
Column-II
(a) Polystyrene
(p) 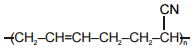
(b) Novolac
(q) 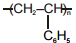
(c) Buna-N
(r) 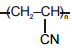
(d) Acrilan
(s) 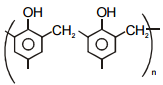
(a)
(b)
(c)
(d)
1.
(s)
(r)
(q)
(p)
2.
(r)
(p)
(s)
(q)
3.
(p)
(q)
(r)
(s)
4.
(q)
(s)
(p)
(r)
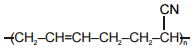
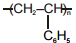
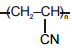

The polymer that can't be prepared by condensation polymerization is :
1. Dacron
2. Nylon-6
3. Glyptal
4. PTFE
The natural polymer among the following options is:
1. Poly(Butadiene-styrene)
2. Polybutadiene
3. Poly(Butadiene-acrylonitrile)
4. Cis-1,4-polyisoprene
On the basis of mode of formation, polymers can be
classified as:
1. Addition polymers only.
2. Condensation polymers only.
3. Co-polymers.
4. Both addition and condensation polymers.
Low-density polythene is prepared by -
1. Free-radical polymerization.
2. Cationic polymerization.
3. Anionic polymerization.
4. Ziegler-Natta polymerization.
Which of the following is a synthetic polymer?
1. Phenol-formaldehyde resin.
2. Proteins.
3. Polysaccharides.
4. Natural rubber.
Teflon, polystyrene, and neoprene are all -
1. Monomers.
2. Co-polymers.
3. Homopolymers.
4. Condensation polymers.
PVC as obtained after polymerization is -
1. Soft and pliable.
2. Hard, brittle, and transparent.
3. Hard and infusible.
4. None of the above.
The compound used in the manufacture of terylene is:
1. Ethylene
2. Vinyl chloride
3. Ethylene glycol
4. Adipic acid
Polymer that is used to make nonstick cookware is:
1. PVC
2. Polystyrene
3. Polythene (terephthalate)
4. Polytetrafluoroethylene


