In the chemical reactions,

the compounds 'A' and 'B', respectively, are:
1. Nitrobenzene and chlorobenzene
2. Nitrobenzene and fluorobenzene
3. Phenol and benzene
4. Benzene diazonium chloride and fluorobenzene

The product formed in the below mentioned reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Alkyl isocyanide on hydrolysis produces:
| 1. | Primary Amine | 2. | Tertiary Amine |
| 3. | Alcohol | 4. | Aldehyde |
The final compound 'B' in the below reaction is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The final product C in the below mentioned reaction is:

| 1. | 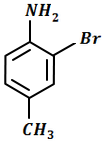 |
2. |  |
| 3. |  |
4. |  |
Mark the correct increasing order of basic strength of given molecules:
 |
 |
 |
| (I) | (II) | (III) |
1. III < I < II
2. III < II < I
3. II < I < III
4. II < III < I
A nitro-compound among the following that does not react with nitrous acid is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
A nitrogen-containing aromatic compound A reacts with Sn/HCl, followed by HNO2 to give an unstable compound B. On treatment with phenol, B forms a beautiful coloured compound C with the molecular formula C12H10N2O. The structure of compound A is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The correct statement regarding the basicity of arylamines is:
| 1. | Arylamines are generally more basic than alkylamines because the lone-pair electrons of nitrogen are not delocalized by interaction with the aromatic ring π-electron system. |
| 2. | Arylamines are generally more basic than alkylamines because of the aryl group +I effect. |
| 3. | Arylamines are generally more basic than alkylamines because the nitrogen atom in arylamines is sp-hybridized. |
| 4. | Arylamines are generally less basic than alkylamines because the nitrogen lone-pair electrons are delocalized by interaction with the aromatic ring π-electron system. |
Mark the reaction that does not yield aniline from the given options:
| 1. | Hydrolysis of phenyl isocyanide with an acidic solution |
| 2. | Degradation of benzamide with bromine in alkaline solution |
| 3. | Reduction of nitrobenzene with H2/Pd in ethanol |
| 4. | Treatment of Potassium salt of phthalimide with chlorobenzene followed by hydrolysis in an aqueous NaOH solution |




