An organic compound 'A' on treatment with NH3 gives 'B', which on heating gives 'C'. 'C',
when treated with Br2 in the presence of KOH produces ethylamine. Compound 'A' is:
1.
CH3CH2COOH
2.
CH3COOH
3.
CH3CH2CH2COOH
4.

when treated with Br2 in the presence of KOH produces ethylamine. Compound 'A' is:

Which of the following compounds is most basic?
| 1. |  |
2. |  |
| 3. |  |
4. |  |
An organic compound () (A), when treated with nitrous acid, gave an alcohol and N2 gas was evolved. (A) on warming with CHCl3 and caustic potash gave (C) which on reduction gave isopropylmethylamine. The structure of (A) is:
| 1. | 2. |  |
|
| 3. |  |
4. |  |
D is N-methyl aniline in the given below sequence of reaction.
\(\text A\xrightarrow{\bf\text{ reduction }}\text B\xrightarrow{\bf\text {CHCl}_3\text{/KOH}}\text C\xrightarrow{\bf\text{ reduction }}\text D\)
structure of A can be:
| 1. |  |
2. |  |
| 3. | CH3NH2 | 4. |  |
The products formed when Aniline reacts with water, and will be, respectively:
| 1. | p-Bromoaniline; p-Chloroaniline |
| 2. | 2,4,6-Tribromoaniline; p-Chloroaniline |
| 3. | 2,4,6-Tribromoaniline; Benzenediazoniumchloride |
| 4. | p-Bromoaniline; Benzenediazoniumchloride |
Aniline in a set of reactions yielded a product D
The structure of the product D would be:
1.
2.
3.
4.
Aniline when diazotized in cold and then treated with dimethyl aniline gives a coloured product. Its structure would be:
| 1. | 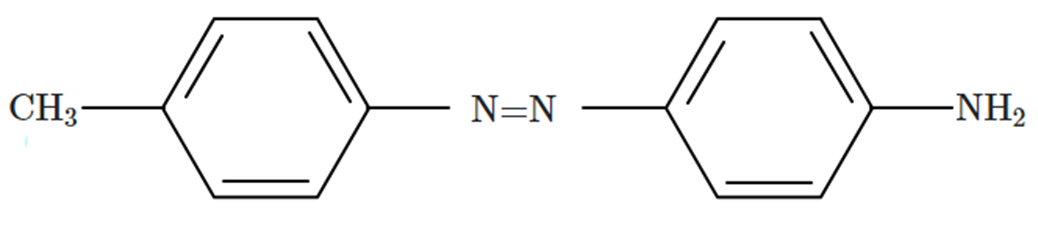 |
| 2. |  |
| 3. |  |
| 4. |  |
Which chemical reaction leads to the formation of isocyanide?
1. Reimer Tiemann reaction.
2. Carbylamine reaction.
3. Hoffmann bromamide reaction.
4. None of the above.
The final product C, obtained in this reaction, would be:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
| 1. | Ethylamine | 2. | Methylamine |
| 3. | Propylamine | 4. | Aniline |




