reacts with benzenesulfonyl chloride to give an insoluble salt in alkali.
The structure of compound (A) will be:
1.
CH3CH2CH2NH2
2.
CH3NHCH2CH3
3.

4.
None of the above

CH3CH2CO-NH2 can be converted into CH3CH2NH2 in the presence of:
1. LiAlH4
2. H2/Pt
3. Sn/HCl
4. NaOBr/OH–
In a set of reactions, propanoic acid yielded a compound (D).
\(CH_3CH_2COOH\ \text{(A)} \xrightarrow[]{SOCl_2}\ \text{(B)} \)
\(\ \xrightarrow[]{NH_3}\ \text{(C)}\ \xrightarrow[Br_2]{KOH}\ \text{(D)}\)
The formula for (D) is:
1.
2.
3.
4.
Aniline in a set of reactions yielded a product D.

The formula of the product D in the above mentioned reaction is
1.
2.
3.
4.
Benzene diazonium chloride on reaction with aniline in the presence of dilute hydrochloric acid gives:
| 1. |  |
2. |  |
| 3. |  |
4. |  |
Which of the following compounds, when reduced with lithium aluminium hydride, produces a secondary amine?
1. Nitroethane
2. Methylisocyanide
3. Acetamide
4. Methyl cyanide
The product B in the below mentioned reaction is:
\(Acetamide \overset{P_2O_5}{\longrightarrow}A\ \overset{4H}{\longrightarrow}\ B\)
1. CH3NH2
2. C2H5NH2
3. CH3CN
4. CH3COONH4
Z in the below reaction sequence will be:
\(CH_3CH_2Cl \)\(\xrightarrow[]{NaCN}\ X\ \)\(\xrightarrow[]{Ni/H_2}\ Y \ \)\(\xrightarrow{Ac_2O}\ Z\)
1. \(\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{NHCOCH}_3
\)
2. \(\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{NH}_2
\)
3. \(\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{CONHCH}_3
\)
4. \(\mathrm{CH}_3 \mathrm{CH}_2 \mathrm{CH}_2 \mathrm{CONHCOCH}_3\)
1. CH3NH2 > (CH3)2NH > (CH3)3N
2. (CH3)2NH > CH3NH2 > (CH3)3N
3. (CH3)3N > CH3NH2 > (CH3)2NH
4. (CH3)3N > CH3NH2 > (CH3)2NH
The structure of C in the below-mentioned reaction is:
| 1. | 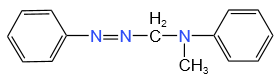 |
| 2. |  |
| 3. |  |
| 4. |  |



