The reaction that does not give benzoic acid as the major product is:
| 1. |  \(\xrightarrow{K_2Cr_2O_7}\) \(\xrightarrow{K_2Cr_2O_7}\) |
2. |  \(\xrightarrow[(ii)H_3O^+]{(i)NaOCl}\) \(\xrightarrow[(ii)H_3O^+]{(i)NaOCl}\) |
| 3. |  \(\xrightarrow{PCC}\) \(\xrightarrow{PCC}\) |
4. |  \(\xrightarrow{KMnO_4/H^+}\) \(\xrightarrow{KMnO_4/H^+}\) |
Subtopic: Aldehydes & Ketones: Preparation & Properties | Carboxylic Acids: Preparation & Properties |
84%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The correct statement among the following about HCOOH is:
1. It is a stronger acid than CH3COOH
2. It reduces Tollen’s reagent
3. It gives CO and H2O on heating with conc.H2SO4
4. All of the above
Subtopic: Carboxylic Acids: Preparation & Properties |
80%
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Match the acids given in Column I with their correct IUPAC names given in Column II and mark the appropriate option:
| Column l (Acids) |
Column ll (IUPAC names) |
| A. Phthalic acid | 1. Hexane-1,6-dioic acid |
| B. Glutaric acid | 2. Benzene-1,2-dicarboxylic acid |
| C. Succinic acid | 3. Pentane-1,5-dioic acid |
| D. Adipic acid | 4. Butane-1,4-dioic acid |
Codes:
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 2 | 1 |
Subtopic: Carboxylic Acids: Preparation & Properties |
79%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Which of the following acids is the strongest?
1. CH3COOH
2. C6H5COOH
3. C6H5OH
4. C2H5COOH
Subtopic: Carboxylic Acids: Preparation & Properties |
73%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.
What is product 'B' in the below mentioned reaction?
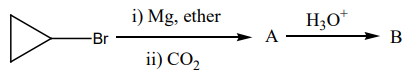
| 1. | 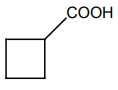 |
2. | 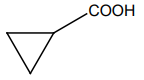 |
| 3. | 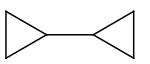 |
4. | 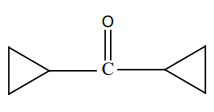 |
Subtopic: Isomers & Reaction Mechanism | Carboxylic Acids: Preparation & Properties |
75%
From NCERT
Please attempt this question first.
Hints
Please attempt this question first.


