The compound on hydrolysis of 50% aqueous sodium hydroxide that produces the corresponding alcohol and acid is:
1.
2.
3.
4.
Consider the following compounds:
| (i) | C6H5COCl | (ii) |  |
| (iii) |  |
(iv) |  |
What is the correct decreasing order of their reactivity towards hydrolysis?
1. (ii) > (iv) > (iii) > (i)
2. (i) > (ii) > (iii) > (iv)
3. (iv) > (ii) > (i) > (iii)
4. (ii) > (iv) > (i) > (iii)
The product formed in aldol condensation is:
| 1. | A \(\beta\)-hydroxy acid |
| 2. | A \(\beta\)-hydroxy aldehyde or a \(\beta\)-hydroxy ketone |
| 3. | An \(\alpha\)-hydroxy aldehyde or ketone |
| 4. | An \(\alpha\)-\(\beta\) unsaturated ester |
A carbonyl compound reacts with hydrogen cyanide to form cyanohydrins, which form a racemic mixture of α-hydroxy acid on hydrolysis. The carbonyl compound is:
1. Acetaldehyde
2. Acetone
3. Diethyl ketone
4. Formaldehyde
The nucleophilic addition reaction will be most favored among the given compounds is:
1.
2.
3.
4.
The self-condensation product of two moles of ethyl acetate in the presence of sodium ethoxide yield is:
1. Ethyl butyrate
2. Acetoacetic ester
3. Methyl acetoacetate
4. Ethyl propionate
The product of the below-mentioned reaction is:
| 1. | 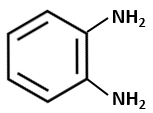 |
2. |  |
| 3. |  |
4. |  |
Among the following acids, which one has the smallest dissociation constant?
| 1. |  |
2. |  |
| 3. |  |
4. | 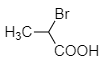 |
The correct statement among the following about HCOOH is:
1. It is a stronger acid than CH3COOH
2. It reduces Tollen’s reagent
3. It gives CO and H2O on heating with conc.H2SO4
4. All of the above
What results from a reaction between concentrated H2SO4 and acetone?
1. Phorone
2. Mesitylene
3. Mesityl oxide
4. Crotonaldehyde



