An organic compound with the molecular formula C9H10O forms a 2,4-DNP derivative, reduces Tollens’ reagent, and undergoes the Cannizzaro reaction. On vigorous oxidation, it gives 1,2-benzenedicarboxylic acid. This organic compound will be:
1.
2-Methyl benzaldehyde
2.
2-Hydroxy benzaldehyde
3.
2-Ethyl benzaldehyde
4.
4-Ethyl benzaldehyde
The below structure is an example of :
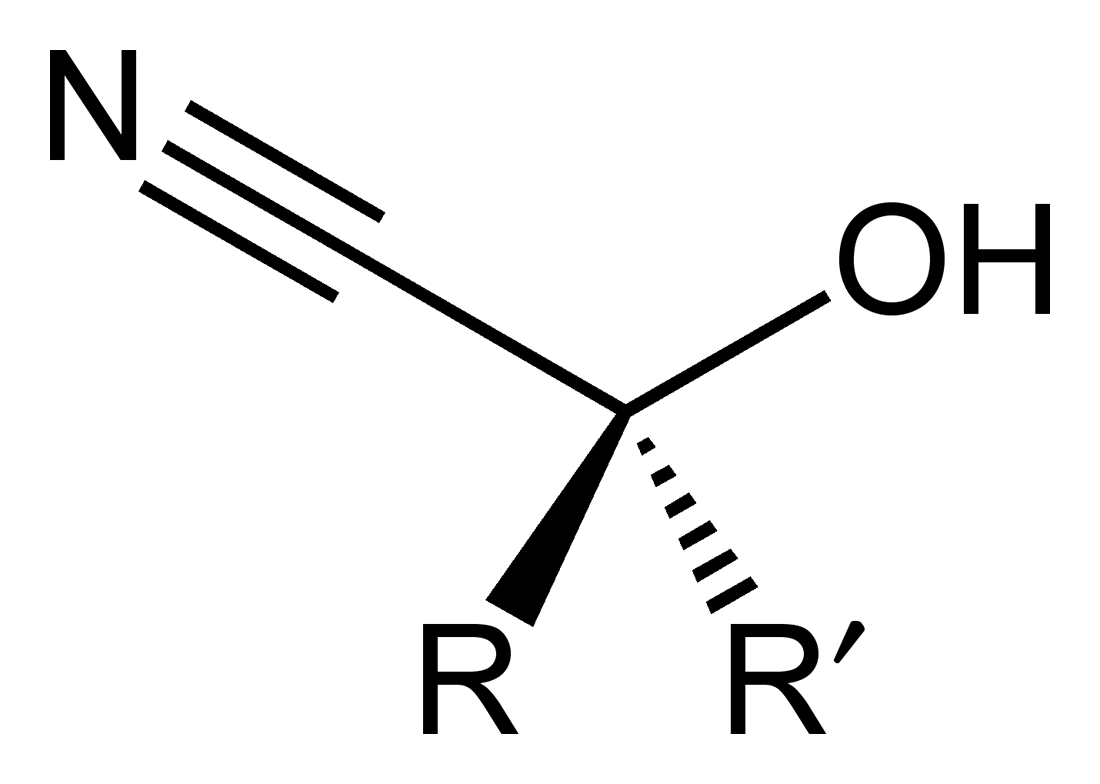
| 1. | Cyanohydrin | 2. | Hemiacetal |
| 3. | Acetal | 4. | Cyanoalcohol |
1. Pentane-2,4-dione
2. Pentane-1,4-dione
3. Pentane-2,2-dione
4. Pentane-3,4-dione
The structure of Hex-2-en-4-ynoic acid is:
| 1. |  |
| 2. |  |
| 3. |  |
| 4. | None of these |
1. Diphenylmethanone
2. Benzophenone
3. Dibenzylmethanone
4. Dibenzylketone
The major product formed when cyclohexanecarbaldehyde reacts with PhMgBr and H3O+ is:
| 1. |  |
2. |  |
| 3. |  |
4. | None of these |
What is the major product of the reaction between cyclohexanecarbaldehyde and Tollens' reagent?
| 1. |  |
2. |  |
| 3. |  |
4. | None of these |
The compounds that undergoes the Cannizzaro reaction are:
(i) Methanal
(ii) 2-Methylpentanal
(iii) Benzaldehyde
(iv) Benzophenone
1. (i)
2. (ii)
3. (i), (iii)
4. (i), (ii), (iii)
Propanal and butanal can produce four different aldol condensation products. The possible structures are:
| 1. |  |
| 2. |  |
| 3. | Both (1) and (2) |
| 4. | None of these |
1. Low electrophilicity in 2,2,6-trimethylcyclohexanone
2. Less steric crowd in 2,2,6-trimethylcyclohexanone
3. More steric crowd in 2,2,6-trimethylcyclohexanone
4. High electrophilicity in 2,2,6-trimethylcyclohexanone


