The hydrogenation of benzoyl chloride in the presence of Pd and BaSO4 gives:
1. Benzyl alcohol
2. Benzaldehyde
3. Benzoic acid
4. Phenol
Oxalic acid on reduction with zinc and gives:
1. Glycol
2. Glyoxal
3. Glyoxalic acid
4. Glycolic acid
X + CH3COOH → Ester + H2O
| 1. | Alcohol | 2. | Pyridine |
| 3. | Secondary amine | 4. | Acid |
Lactic acid on oxidation with alkaline potassium permanganate gives:
1. Propionic acid
2. Cinnamic acid
3. Pyruvic acid
4. Tartaric acid
The compound that can give a positive iodoform test is:
1. 2-Phenylethanol
2. Pentanal
3. 3-Pentanol
4. 1-Phenylethanol
Benzoyl chloride can be prepared from benzoic acid by reacing the former with:
1. Cl2, hv
2. SO2
3. SOCl2
4. Cl2, H2O
When treated with alkali and iodine, what substance will not give iodoform?
| 1. | Acetone | 2. | Ethanol |
| 3. | Diethyl ketone | 4. | isopropyl alcohol |
The compound that responds to Tollen's test is
1.
2.
3.
4.
Concentrated sodium hydroxide and benzaldehyde react to produce:
1. Cinnamic acid
2. Hydrobenzamide
3. Benzyl alcohol
4. Benzophenone
An alkene “A” on reaction with O3 and Zn - H2O gives propanone and ethanal in an equimolar ratio. The addition of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is:
| 1. | 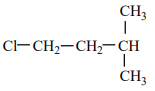 |
2. | 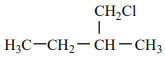 |
| 3. | 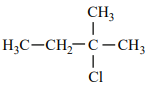 |
4. | 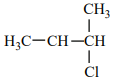 |


