An alkene “A” on reaction with O3 and Zn - H2O gives propanone and ethanal in an equimolar ratio. The addition of HCl to alkene “A” gives “B” as the major product. The structure of product “B” is:
1.
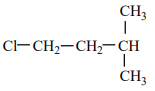
2.
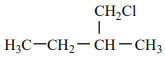
3.
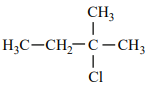
4.
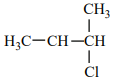
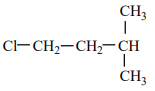
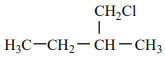
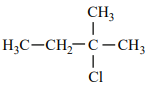
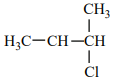
Concentrated sodium hydroxide and benzaldehyde react to produce:
1. Cinnamic acid
2. Hydrobenzamide
3. Benzyl alcohol
4. Benzophenone
The compound that responds to Tollen's test is
1.
2.
3.
4.
When treated with alkali and iodine, what substance will not give iodoform?
| 1. | Acetone | 2. | Ethanol |
| 3. | Diethyl ketone | 4. | isopropyl alcohol |
Benzoyl chloride can be prepared from benzoic acid by reacing the former with:
1. Cl2, hv
2. SO2
3. SOCl2
4. Cl2, H2O
The hydrogenation of benzoyl chloride in the presence of Pd and BaSO4 gives:
1. Benzyl alcohol
2. Benzaldehyde
3. Benzoic acid
4. Phenol
Oxalic acid on reduction with zinc and gives:
1. Glycol
2. Glyoxal
3. Glyoxalic acid
4. Glycolic acid
X + CH3COOH → Ester + H2O
| 1. | Alcohol | 2. | Pyridine |
| 3. | Secondary amine | 4. | Acid |
Lactic acid on oxidation with alkaline potassium permanganate gives:
1. Propionic acid
2. Cinnamic acid
3. Pyruvic acid
4. Tartaric acid
The compound that can give a positive iodoform test is:
1. 2-Phenylethanol
2. Pentanal
3. 3-Pentanol
4. 1-Phenylethanol


