The steam-volatile compound x among the following is:
1. o-Nitrophenol
2. p-Nitrophenol
3. Both 1 and 2
4. None of the above
Ortho nitrophenol is more acidic than ortho methoxy phenol because:
| 1. | The NO2-group increases the electron density in phenol while the methoxy group decreases the electron density in phenol |
| 2. | The nitro-group is an electron-withdrawing group while the methoxy group is an electron-releasing group |
| 3. | The methoxy-group is an electron-withdrawing group while the nitro group is an electron-releasing group |
| 4. | None of the above |
Preparation of ethers by 2 or 3-degree alcohols in an acidic medium is not a suitable method because:
| 1. | In case of secondary or tertiary alcohols, ketone is obtained as a product. |
| 2. | In case of secondary or tertiary alcohols, aldehyde is obtained as a product. |
| 3. | In case of secondary or tertiary alcohols, alkene is obtained as a product. |
| 4. | In case of primary alcohols, alkene is obtained as a product. |
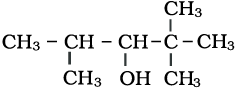
The IUPAC name of the above compound is:
1. 2, 2, 2-Trimethylpentan-3-ol
2. 2, 2, 4-Trimethylpentan-3-ol
3. 2, 2, 3-Trimethylpentan-3-ol
4. 1, 1, 3-Trimethylpentan-3-ol
The IUPAC name of the below compound is:

| 1. | 1-Phenylpropan-2-ol | 2. | 2-Phenylpropan-2-ol |
| 3. | 1-Phenylpropan-1-ol | 4. | 2-Phenylpropan-1-ol |
The IUPAC name of the below compound is:

1. 3,4-Dimethylhexane –1,3,5-triol
2. 3,5-Dimethylhexane –1,1,5-triol
3. 3,5-Dimethylhexane –1,3,5-triol
4. 3,1-Dimethylhexane –1,3,5-triol

The IUPAC name of the above compound is:
| 1. | 4 – Ethoxypropane | 2. | 3 – Ethoxypropane |
| 3. | 1 – Ethoxypropane | 4. | 2 – Ethoxypropane |
The IUPAC name of the following compound is:

1. 1-Ethoxy-3-methylpentane
2. 3-Ethoxy-1-methylpentane
3. 2-Ethoxy-3-methylpentane
4. 3-Ethoxy-2-methylpentane

The structure and name of intermediate compound is -
| 1. |  Cumene hydroperoxide |
| 2. |  Cumene hydroperoxide |
| 3. |  Cumene hydroperoxide |
| 4. | None of these |
The IUPAC name of the following compound is:

1. Cyclopent-1-en-3-ol
2. Cyclopent-3-en-1-ol
3. Cyclopent-2-en-1-ol
4. Cyclopent-1-en-2-ol


