Select Chapter Topics:
Match the items in Column I and Column II.
Column l
Column ll
A. SN1 reaction
1. Vic-dibromide
B. Elimination of HX from an alkyl halide
2. Chlorobromocarbons
C. Bromination of alkenes
3. Racemisation
D. Chemicals in fire extinguisher
4. Saytzeff rule
Codes:
A
B
C
D
1.
2
3
4
1
2.
3
4
1
2
3.
1
4
3
2
4.
4
1
3
2
Subtopic: Chemical Properties |
86%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the structures of compounds given in Column I with the classes of compounds given in Column II.
| Column I | Column II | ||
| A. |  |
1. | Aryl halide |
| B. |  |
2. | Alkyl halide |
| C. |  |
3. | Vinyl halide |
| D. |  |
4. | Allylic halide |
Codes:
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 3 | 4 | 1 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Introduction and Physical Properties |
84%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the reactions given in Column I with the types of reactions given in Column II and mark the appropriate option.
| Column I | Column II | ||
| A. |  |
1. | Nucleophilic aromatic substitution reaction |
| B. | 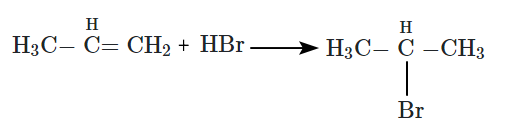 |
2. | Electrophilic aromatic substitution |
| C. |  |
3. | Saytzeff elimination |
| D |  |
4. | Electrophilic addition |
| 5. | Nucleophilic substitution reaction |
Codes:
| A | B | C | D | |
| 1. | 2 | 4 | 5 | 1 |
| 2. | 3 | 1 | 5 | 2 |
| 3. | 5 | 4 | 3 | 2 |
| 4. | 4 | 5 | 3 | 2 |
Subtopic: Chemical Properties |
74%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Match the structures given in Column I with the names in Column II.
| Column I | Column II | ||
| A. |  |
1. | 4-Bromopent-2-ene |
| B. |  |
2. | 4-Bromo-3-methylpent-2-ene |
| C. |  |
3. | 1-Bromobut-2-ene |
| D. |  |
4. | 1-Bromo-2-methylpent-2-ene |
Codes:
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Introduction and Physical Properties |
89%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The correct match of column I (Reactions) with column II (Name of reactions) is:
| Column I | Column II | ||
| A. |  |
1. | Fittig reaction |
| B. |  |
2. | Wurtz-Fittig reaction |
| C. |  |
3. | Finkelstein reaction |
| D. |  |
4. | Sandmeyer reaction |
Codes:
| A | B | C | D | |
| 1. | 2 | 1 | 4 | 3 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Subtopic: Chemical Properties |
90%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | Phosphorus chlorides (tri and Penta) are preferred over thionyl chloride for the preparation of alkyl chlorides from alcohols. |
| Reason (R): | Phosphorus chlorides give pure alkyl halides. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Subtopic: Chemical Properties |
69%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | The boiling points of alkyl halides decrease in the order RI> RBr > RCl > RF |
| Reason (R): | The boiling points of alkyl chlorides, bromides, and iodides are considerably higher than those of the hydrocarbons of comparable molecular mass. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Introduction and Physical Properties |
59%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | KCN reacts with methyl chloride to give methyl isocyanide. |
| Reason (R): | \(CN^-\) is an ambident nucleophile. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Chemical Properties |
72%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | The presence of a nitro group at ortho or para position increases the reactivity of haloarenes towards nucleophilic substitution. |
| Reason (R): | The nitro group, being an electron-withdrawing group, decreases the electron density over the benzene ring. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Haloarenes: Directive Influence of Halogen |
81%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select the correct option based on statements below:
| Assertion (A): | In monohaloarenes, further electrophilic substitution occurs at ortho and para positions. |
| Reason (R): | The halogen atom is a ring deactivator. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Haloarenes: Directive Influence of Halogen |
53%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch


