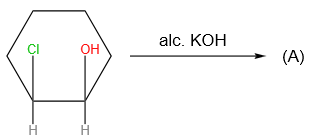
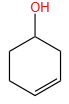
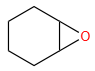
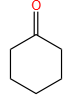
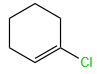
Sandmeyer's reaction described by among the following is:
1.

2.

3.

4.


| 1. |  |
| 2. |  |
| 3. |  |
| 4. |  |
Which of the following molecules can readily undergo the SN2 mechanism?
| 1. | \(\left(\mathrm{CH}_3\right)_3 \mathrm{C}-\mathrm{Cl} \) | 2. | \(\mathrm{CH}_2=\mathrm{CH}-\mathrm{Cl} \) |
| 3. | \(\mathrm{CH}_2=\mathrm{CH}-\mathrm{CH}_2-\mathrm{CH}_2 \mathrm{Cl}\) | 4. |  |
The X and Y in the below mentioned reaction are
| X | Y | |
| 1. | Dilute aqueous NaOH, 20 oC | HBr/Acetic acid, 20 oC |
| 2. | Concentrated alcoholic NaOH, 80 oC | HBr/Acetic acid, 20 oC |
| 3. | Diluted aqueous NaOH, 20 oC, | Br2 / CHCl3 , 0 oC |
| 4. | Concentrated alcoholic NaOH, 80 ° C | Br2/CHCl3, 0 oC |
What is the total number of chiral products produced by monochlorination of 2-methylbutane ?
1. 2
2. 4
3. 6
4. 8
How is m-Bromotoluene prepared?
| 1. | Bromination of toluene |
| 2. | Friedel Craft’s reaction of bromobenzene with CH3Cl |
| 3. | Bromination of nitrobenzene and subsequent replacement of –NO2 group with an ethyl group |
| 4. | Bromination of Aceto–p–toluidine followed by hydrolysis and deamination |
Products (A) and (B) in the given below reactions, respectively, are:

| (A) | (B) | |
| 1. |  |
 |
| 2. |  |
 |
| 3. |  |
 |
| 4. | None of the above | |
When vinyl chloride is passed through an alcoholic KOH solution, which of the following is correct?
1. It gets dissolved.
2. It forms vinyl alcohol
3. It forms acetylene
4. It has no action
The product (C) in the below-mentioned reaction is:
\(CH_{3}CH_{2}CH_{2}Br \ \xrightarrow[]{\textbf{KOH(alc.)}} \)
\(\ (A) \ \xrightarrow[]{\textbf{HBr}} \ (B) \ \xrightarrow[]{\textbf{KOH(aq}} \ (C)\)
| 1. | Propene | 2. | Propyne |
| 3. | Propan-1-ol | 4. | Propan-2-ol |