The shape of Fe(CO)5 is:
1.
Octahedral
2.
Square planar
3.
Trigonal bipyramidal
4.
Square pyramidal
Which one of the following is expected to exhibit optical isomerism?
(en = ethylenediamine)
1. cis-[Pt(NH3)2Cl2]
2. cis-[Co(en)2Cl2]
3. trans−[Co(en)2Cl2]
4. trans−[Pt(NH3)2Cl2]
Mg is present in:
| 1. | Chlorophyll | 2. | Haemoglobin |
| 3. | Vitamin-B12 | 4. | Vitamin-B |
Number of isomers of complex are:
| 1. | 2 | 2. | 3 |
| 3. | 4 | 4. | 5 |
The coordination compound that will give four isomers is:
1. [Fe(en)3]Cl3
2. [Co(en)2Cl2]Cl
3. [Fe(PPh3)3NH3ClBr]Cl
4. [Co(PPh3)3Cl]Cl3
The IUPAC name of will be:
1. Triamminebromochloronitrocobaltate (III)
2. Triamminebromochloronitrocobalt (III)
3. Triamminebromonitrochlorocobalt (III)
4. Triamminenitrobromochlorocobalt (III)
In [Cr(NH3)6](Br)3, number of unpaired electrons in Cr is:
| 1. | 4 | 2. | 3 |
| 3. | 1 | 4. | 2 |
Anticancer agent among the following is:
| 1. |  |
2. | 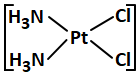 |
| 3. |  |
4. |  |
In an octahedral structure, the pair of d orbitals involved in d2sp3 hybridization is:
1.
2.
3.
4.
The strong field nature of CN⁻ as a ligand can be attributed to:
1. It is a pseudohalide.
2. It can accept electrons from metal species.
3. It forms high-spin complexes with metal species.
4. It carries a negative charge.


