Which of the following statement about collodion is correct?
1.
4% solution of nitro-cellulose in a mixture of ethyl alcohol and ether.
2.
14% solution nitrate in a mixture of ether and ethanol.
3.
20 % solution nitrate in a mixture of propanol and diethyl ether.
4.
None of the above.
The correct curve for Freundlich's adsorption isotherm is:
| 1. | 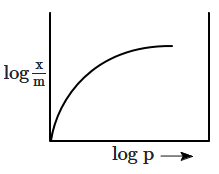 |
2. |  |
| 3. |  |
4. |  |
Match the types of colloidal systems given in Column I with the names given in Column II.
| Column I | Column II |
| A. Solid in Liquid | 1. Foam |
| B. Liquid in solid | 2. Sol |
| C. Liquid in Liquid | 3. Gel |
| D. Gas in Liquid | 4. Emulsion |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Select the correct option based on statements below:
| Assertion (A): | An ordinary filter paper impregnated with collodion solution stops the flow of colloidal particles. |
| Reason (R): | The pore size of the filter paper becomes larger than the size of the colloidal particle. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Select the correct option based on statements below:
| Assertion (A): | Colloidal solutions do not show Brownian motion. |
| Reason (R): | Brownian motion is responsible for the stability of sols. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Select the correct option based on statements below:
| Assertion (A): | The coagulation power of Al3+ is more than Na+. |
| Reason (R): | Greater the valency of the flocculating ion added, the greater is its power to cause precipitation (Hardy-Schulze rule) |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Select the correct option based on statements below:
| Assertion (A): | Detergents with low CMC are more economical to use. |
| Reason (R): | The cleansing action of detergents involves the formation of micelles. These are formed when the concentration of detergents becomes equal to CMC. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Match the items in Column I and Column II.
| Column I | Column II |
| A. Dialysis | 1. Cleansing action of soap |
| B. Peptization | 2. Coagulation |
| C. Emulsification | 3. Colloidal sol formation |
| D. Electrophoresis | 4. Purification |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 3 | 1 | 2 |
Which one of the following is the correct option?
|
1. |
Solid sol |
- |
Smoke |
|
2. |
Emulsion |
- |
butter |
|
3. |
Foam |
- |
Mist |
|
4. |
Whipped cream |
- |
sol |
Among the following surfactant molecules, the surfactant with the lowest CMC in aqueous solution at ambient conditions is:
1. CH3(CH2)15N+(CH3)3Br-
2. CH3(CH2)11OSO3- Na+
3. CH3(CH2)6COO-Na+
4. CH3(CH2)11N+(CH3)3Br-


