How can alum help in water purification?
1.
Forming Si complex with clay particles.
2.
The sulphate part, which combines with the dirt and removes it.
3.
Aluminium which coagulates the mud particles.
4.
Making mud water soluble.
Measuring Zeta potential is useful in determining which of the following property of a colloidal solution:
1. Solubility.
2. Stability of the colloidal particles.
3. Size of the colloidal particles.
4. Viscosity.
Brownian motion of colloidal particles is caused by:
| 1. | Temperature fluctuations within the liquid phase. |
| 2. | Attraction and repulsion between charges on the colloidal particles. |
| 3. | Impact of the molecules of the dispersion medium on the colloidal particles. |
| 4. | None of the above. |
Select the correct option based on statements below:
| Assertion (A): | One part of micelles is lyophilic in nature. |
| Reason (R): | Micelles have a lyophillic COO group. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
Select the correct option based on statements below:
| Assertion (A): | A hydrophilic sol is more stable than a hydrophobic sol. |
| Reason (R): | When compared to hydrophobic sol, colloidal particles in hydrophilic sol are heavily hydrated. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |
The correct curve for Freundlich's adsorption isotherm is:
| 1. | 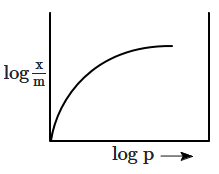 |
2. |  |
| 3. |  |
4. |  |
The coagulating power of electrolytes with ions Na+, Al3+, and Ba2+for arsenic sulphide sol increases in the order :
1.
2.
3.
4.
The method of formation of the solution is given in Column I. Match it with the type of solution given in Column II.
| Column I | Column II |
| (A) Sulphur vapours passed through cold water | 1. Normal electrolyte solution |
| (B) Soap mixed with water above critical micelle concentration | 2. Molecular colloids |
| (C) White of egg whipped with water | 3. Associated colloid |
| (D) Soap mixed with water below critical micelle concentration | 4. Macromolecular colloids |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 4 | 1 | 3 | 2 |
Match the statement given in Column I with the phenomenon given in Column II.
| Column I | Column II |
| A. Dispersion medium moves in an electric field | 1. Osmosis |
| B. Solvent molecules pass through a semi-permeable membrane towards the solvent side | 2. Electrophoresis |
| C. Movement of charged colloidal particles under the influence of applied electric potential towards oppositely charged electrodes | 3. Electroosmosis |
| D. Solvent molecules pass through semi-permeable membranes towards the solution side | 4. Reverse osmosis |
Codes
| A | B | C | D | |
| 1. | 2 | 3 | 4 | 1 |
| 2. | 1 | 2 | 3 | 4 |
| 3. | 3 | 4 | 2 | 1 |
| 4. | 4 | 1 | 3 | 2 |
The equation that represents a Freundlich adsorption isotherm is:
1.
2.
3.
4.


