The half-life for a zero-order reaction having 0.02 M initial concentration of reactant is 100 s. The rate constant (in mol L–1 s–1) for the reaction is:
1.
2.
3.
4.
What does ZAB represent in the collision theory of chemical reactions?
| 1. | The fraction of molecules with energies greater than Ea |
| 2. | The collision frequency of reactants, A and B |
| 3. | Steric factor |
| 4. | The fraction of molecules with energies equal to Ea |
For a reaction AB, enthalpy of reaction is and enthalpy of activation is . The correct potential energy profile for the reaction is:
| 1. | 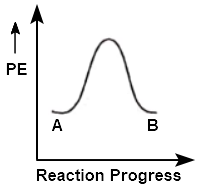 |
2. |  |
| 3. |  |
4. |  |
The slope of Arrhenius Plot (ln k v/s ) of the first-order reaction is . The value of Ea of the reaction is:
[Given R = 8.314 JK–1 mol–1]
| 1. | 166 kJ mol–1 | 2. | –83 kJ mol–1 |
| 3. | 41.5 kJ mol–1 | 4. | 83.0 kJ mol–1 |
An increase in the concentration of the reactants of a reaction leads to a change in:
| 1. | Heat of reaction | 2. | Threshold energy |
| 3. | Collision frequency | 4. | Activation energy |
The rate constant for a first order reaction is . The time required to reduce 2.0 g of the reactant to 0.2 g is:
| 1. | 200 s | 2. | 500 s |
| 3. | 1000 s | 4. | 100 s |

The y and x axes for zero and first-order reactions, respectively are
| 1. | zero order (y=rate and x=concentration), first order (y=rate and x=t1/2) |
| 2. | zero order (y=concentration and x=time), first order (y=t1/2 and x = concentration) |
| 3. | zero order (y=concentration and x= time), first order (y=rate constant and x= concentration) |
| 4. | zero order (y=rate and x=concentration), first order (y=t1/2 and x = concentration) |
For a first-order reaction A \(\rightarrow\) Products, initial concentration of A is 0.1 M, which becomes 0.001 M after 5 minutes. Rate constant for the reaction in min-1 is
| 1. | 0.2303 | 2. | 1.3818 |
| 3. | 0.9212 | 4. | 0.4606 |
For the reaction, 2A → B, rates= k[A]2. If the concentration of reactant is doubled, then the:
| (a) | rate of reaction will be doubled. |
| (b) | rate constant will remain unchanged, however rate of reaction is directly proportional to the rate constant. |
| (c) | rate constant will change since the rate of reaction and rate constant are directly proportional to each other. |
| (d) | rate of reaction will increase by four times. |
Identify the set of correct statements & choose the correct answer from the options given below:
| 1. | (a) and (c) only | 2. | (a) and (b) only |
| 3. | (b) and (d) only | 4. | (c) and (d) only |
The plot of ln k vs \({1 \over T}\) for the following reaction
\(2N_2O_5(g) \rightarrow 4NO_2 (g) + O_2(g) \) gives a straight line with the slope of the line equal to \(-1.0 \times 10^4 K \).
The activation energy for the reaction in J mol–1 is:
(Given R = 8.3 J K–1 mol–1)
| 1. | \(4.0 \times 10^2 \) | 2. | \(4.0 \times 10^{-2} \) |
| 3. | \(8.3 \times 10^{-4} \) | 4. | \(8.3 \times 10^4 \) |


