At high pressure the following reaction is zero order-

The correct statements among the following is:
(a)
Rate of reaction = Rate constant
(b)
Rate of the reaction depends on the concentration of ammonia
(c)
Rate of decomposition of ammonia will remain constant until ammonia disappears completely
(d)
Further increase in pressure will change the rate of reaction
1.
(a, b, c)
2.
(b, c, d)
3.
(a, c, d)
4.
(a, b, d)

During decomposition of an activated complex
| (a) | Energy is always released. |
| (b) | Energy is always absorbed. |
| (c) | Energy does not change. |
| (d) | Reactants may be formed. |
The correct choice among the given is -
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
The correct statements among following about Maxwell, Boltzmann distribution of energy is -
| (a) | The fraction of molecules with the most probable kinetic energy decreases at higher temperatures |
| (b) | The fraction of molecules with the most probable kinetic energy increases at higher temperatures |
| (c) | Most probable kinetic energy increases at higher temperatures |
| (d) | Most probable kinetic energy decreases at higher temperatures |
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)
The correct statements among the following regarding Maxwell, Boltzmann distribution curve of energy is -
| a. | The area under the curve must not change with an increase in temperature. |
| b. | The area under the curve increases with increase in temperature. |
| c. | Area under the curve decreases with increase in temperature. |
| d. | With an increase in temperature curve broadens and shifts to the right-hand side. |
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
The correct statements among the following regarding the Arrhenius equation is/are -
| (a) | Rate of a reaction increases with an increase in temperature |
| (b) | Rate of a reaction increases with a decrease in activation energy |
| (c) | Rate constant decreases exponentially with an increase in temperature |
| (d) | Rate of reaction decreases with a decrease in activation energy |
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
The correct statements among the following is:
| (a) | Catalyst provides an alternative pathway to the reaction mechanism |
| (b) | Catalyst raises the activation energy |
| (c) | A catalyst lowers the activation energy |
| (d) | Catalyst alters enthalpy change of the reaction |
| 1. | (a, b) | 2. | (b, c) |
| 3. | (c, d) | 4. | (a, c) |
The graphs that represent a zero-order reaction are:
| (a) | 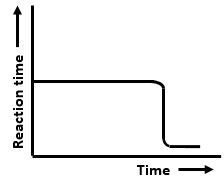 |
(b) |  |
| (c) |  |
(d) |  |
| 1. | (a, b) | 2. | (b, c) |
| 3. | (c, d) | 4. | (a, d) |
The correct graphical representation of first-order reaction is:
| (a) |  |
(b) |  |
| (c) |  |
(d) |  |
| 1. | (a) and (b) | 2. | (b) and (c) |
| 3. | (c) and (d) | 4. | (a) and (d) |
Match the graph given in Column I with the order of reaction given in Column II.
More than one item in Column I may be linked to the same item in Column II:
| Column I | Column II | ||
| (i) |  |
(a) | 1st order |
| (ii) |  |
(b) | Zero order |
| (iii) |  |
||
| (iv) |  |
||
| (i) | (ii) | (iii) | (iv) | |
| 1. | (a) | (b) | (a) | (b) |
| 2. | (a) | (b) | (b) | (a) |
| 3. | (a) | (a) | (b) | (b) |
| 4. | (b) | (b) | (a) | (a) |
| Column I | Column II | ||
| A. | Catalyst alters the rate of reaction | 1. | Proper orientation is not there always |
| B. | 2. | By lowering the activation energy | |
| C. | Energetically favorable reactions are sometimes slow | 3. | Total probability is one |
| D. | Area under the Maxwell-Boltzmann curve is constant | 4. | Refers to the fraction of molecules with energy equal to or greater than the activation energy |
Codes:
| A | B | C | D | |
| 1. | 2 | 4 | 1 | 3 |
| 2. | 3 | 1 | 4 | 2 |
| 3. | 1 | 4 | 3 | 2 |
| 4. | 3 | 4 | 1 | 2 |


