The correct statement regarding the comparison of staggered and eclipsed conformations of ethane is:
1.
The eclipsed conformation of ethane is more stable than staggered conformation because eclipsed conformation has no torsional strain
2.
The eclipsed conformation of ethane is more stable than staggered conformation even though the eclipsed conformation has a torsional strain
3.
The staggered conformation of ethane is more stable than the eclipsed conformation because staggered conformation has no torsional strain
4.
The staggered conformation of ethane is less stable than eclipsed conformation because staggered conformation has torsional strain
The correct order of strengths of the carboxylic acids :-
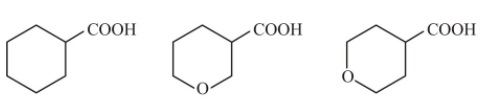
(a) I>II>III
(b) II>III>I
(c) III>II>I
(d) lI>I>IlI
Which among the given molecules can exhibit tautomerisrn?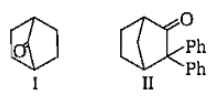
1. III Only
2. Both I and III
3. Both Iand II
4. Both II and III
The correct order of acidity among the following is:
1. CH2=CH2 >CH≡CH > CH3C≡CH > CH3-CH3
2. CH≡CH > CH3-C≡CH > CH2=CH2 >CH3-CH3
3. CH≡CH > CH2=CH2 > CH3-C≡CH > CH3-CH3
4. CH3-CH3 > CH2=CH2 > CH3-C≡CH > CH≡CH
With respect to the conformers of ethane, which of the following statements is true?
(1) Bond angle remains same but bond length changes
(2) Bond angle changes but bond length remains same
(3) Both bond angle and bond length change
(4) Both bond angles and bond length remain same
The IUPAC name of the above mentioned compound is-
1. 3-Ket-2-methylhex-4-enal
2. 5-Formylhex-2-en-3-one
3. 5-Methyl-4-oxohex-2-en-5-al
4. 3-Keto-2-methylhex-4-enal
The most suitable method used for the separation of 1:1 mixture of ortho and para-nitrophenols is:
| 1. | Chromatography | 2. | Crystallization |
| 3. | Steam distillation | 4. | Sublimation |
The IUPAC name of the compound 
1. 5-formylhex-2-en-3-one
2. 5-methyl-4-2-en-5-el
3. 3-keto-2-methylhex-5-enal
4. 3-keto-2-methylhex-4-enal
The correct statement regarding electrophile is :
| 1. | Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile |
| 2. | Electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile |
| 3. | Electrophiles can be either neutral or positively charged species and can form a bond accepting a pair of electrons from a nucleophile |
| 4. | Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile |
In pyrrole
the electron density is maximum on:
1. 2 and 3
2. 3 and 4
3. 2 and 4
4. 2 and 5




