The optically active compound among the following is-
1.
Glycerine
2.
Acetaldehyde
3.
Glyceraldehyde
4.
Acetone
Glucose and fructose are-
| 1. | Chain isomers | 2. | Position isomers |
| 3. | Functional isomers | 4. | Optical isomers |
The optically active compound among the following is-
1. Isobutyric acid
2. beta-Chloropropionic acid
3. Propionic acid
4. alpha-Chloropropionic acid
The correct order of acidity among the following is:
1. CH2=CH2 >CH≡CH > CH3C≡CH > CH3-CH3
2. CH≡CH > CH3-C≡CH > CH2=CH2 >CH3-CH3
3. CH≡CH > CH2=CH2 > CH3-C≡CH > CH3-CH3
4. CH3-CH3 > CH2=CH2 > CH3-C≡CH > CH≡CH
The compound that gives the most stable carbonium ion after C- Cl bond ionisation among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |
The enolic form of ethyl acetoacetate is given below. The number of sigma and pi bonds in the enolic form of ethyl acetoacetate are -
1. 18 sigma bonds and 2 pi-bonds
2. 16 sigma bonds and 1 pi-bond
3. 9 sigma bonds and 2 pi-bonds
4. 9 sigma bonds and 1 pi-bond
The incorrect statement among the following for a nucleophile is:
1. Nucleophile is a Lewis acid
2. Ammonia is a nucleophile
3. Nucleophiles attack low electron density sites
4. Nucleophiles are not electron seeking
In Duma's method of estimation of nitrogen, 0.35 g of an organic compound gave 55 ml of nitrogen collected at 300 K temperature and 715 mm pressure. The percentage composition of nitrogen in the compound would be:
(Aqueous tension at 300 K = 15 mm)
1. 16.45
2. 27.45
3. 44.45
4. 35.45
The most acidic compound among the following is-
| 1. |  |
2. |  |
| 3. |  |
4. |  |
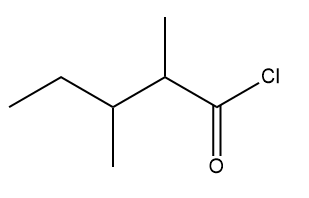
The IUPAC name of the above mentioned compound is -
1. 3, 4-Dimethylpentanoyl chloride
2. 1-Chloro-1-oxo-2,3-dimethylpentane
3. 2-Ethyl-3-methylbutanoylchloride
4. 2, 3-Dimethylpentanoyl chloride



