0.2 g of an organic compound on complete combustion produces 0.44 g of CO2. The percentage of carbon is -
1. 50%
2. 60%
3. 70%
4. 80%
The IUPAC name of the above mentioned compound is-
1. 3-Ket-2-methylhex-4-enal
2. 5-Formylhex-2-en-3-one
3. 5-Methyl-4-oxohex-2-en-5-al
4. 3-Keto-2-methylhex-4-enal
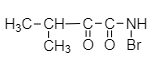
The IUPAC name of the above-mentioned compound is -
1. (N-Bromo)-3-methyl-2-oxobutanamide
2. (N-Bromo)-2-oxo-4-methylbutanamide
3. (N-Bromo)-1,2-dioxo-3-methylbutanamine carboxamide
4. (N-Bromo)-1-oxo-2-methylpropane
The colour of the solution that gets formed by mixing sodium nitroprusside to an alkaline solution of sulfide ions, is-
| 1. | Red | 2. | Blue |
| 3. | Brown | 4. | Purple |
In Kjeldahl's method, the nitrogen present is estimated as-
1. N2
2. NH3
3. NO2
4 None of the above
In Kjeldahl's method of estimation of nitrogen, K2SO4 acts as:
| 1. | An oxidizing agent | 2. | A catalytic agent |
| 3. | A hydrolyzing agent | 4. | A boiling point elevator |
The Prussian blue colour obtained during the test of nitrogen by Lassaigne's test is due to the formation of-
| 1. | Fe4[Fe(CN)6]3 | 2. | Na3[Fe(CN)6] |
| 3. | Fe(CN)3 | 4. | Na4[Fe(CN)5NOS] |
Soda extract is prepared by-
1. Fusing soda and mixture of hydrocarbons, and then extracted with water
2. Dissolving NaHCO3 and mixture of hydrocarbons in dil. HCl
3. Boiling Na2CO3 and mixture of hydrocarbons in dil. HCl
4. Boiling Na2CO3 and mixture of hydrocarbons in distilled water
The most stable carbocation among the following is:
| 1. |  |
2. |  |
| 3. | 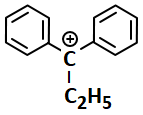 |
4. |  |
The arrangement in decreasing order of stability of H3, H5,(CH3)2H and (CH3)3 free radicals is-
1. H3 > H5 > (CH3)2H > (CH3)3
2. (CH3)3 > (CH3)2H > H5 > H3
3. H5 > H3 > (CH3)2H > (CH3)3
4. (CH3)3 > (CH3)2H > H3 >H5



