In Carius method of estimation of halogen, 0.15 g of an organic compound gave 0.12 g of AgBr.
The percentage of bromine in the compound is-
1. 39.05 %
2. 42.42 %
3. 34.04%
4. 65.12 %
In sulphur estimation, 0.157 g of an organic compound gave 0.4813 g of barium sulphate. The percentage of sulphur in the compound is-
1. 39.10 %
2. 48.13 %
3. 42.10 %
4. 52. 43 %
During estimation of nitrogen present in an organic compound, the ammonia evolved from 0.5 g of the compound in Kjeldahl’s estimation of nitrogen, neutralized 10 mL of 1 M H2SO4. The percentage of nitrogen in the compound is-
1. 46.0 %
2. 51.0%
3. 56.0%
4. 49.0%
In Dumas’ method for estimation of nitrogen, 0.3g of an organic compound gave 50mL of nitrogen collected at 300K temperature and 715mm pressure. The percentage composition of nitrogen in the compound is-
(Aqueous tension at 300K=15 mm)
1. 21.43%
2. 25.13%
3. 19.46%
4. 17.46%
On complete combustion, 0.246 g of an organic compound gave 0.198g of carbon dioxide and 0.1014g of water.
The percentage composition of carbon and hydrogen in the compound are respectively-
1. 21.95 %; 4.58 %
2. 33.46 %; 3.14 %
3. 42.15 %; 2.61 %
4. 25.24 %; 3.98 %
The correct order of stability of following carbocation is-
| 1. | b > a > c | 2. | a > c > b |
| 3. | a > b > c | 4. | c > b > a |
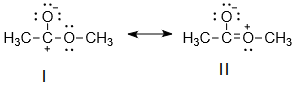
Consider the resonance structure of CH3COOCH3 as follows:
The correct statement among the following is-
| 1. | I and II cannot be the major contributors to the real structure of CH3COOCH3 |
| 2. | I and II can be the major contributors to the real structure of CH3COOCH3 |
| 3. | I is more stable than II. |
| 4. | None of the above |
Consider the possible resonating structures of CH2=CH–CHO is as follows:
The correct sequence of stability is-
1. II > I > III
2. III > I > II
3. I > III > II
4. I > II > III
The correct resonance structures of CH3COO- among the following are:
| 1. |  |
| 2. | |
| 3. | |
| 4. |  |
Consider the following compound:
Carbon with minimum impact of negative inductive effect of Br is -
1. C1
2. C2
3. C3
4. All have same inductive effect