The compound that is most difficult to protonate is:
1.

2.

3.

4.





Paper chromatography is an example of-
1. Partition chromatography
2. Thin layer chromatography
3. Column chromatography
4. Adsorption chromatography
A liquid compound (X) can be purified by steam distillation only if it is:
1. Steam volatile, immiscible with water.
2. Not steam volatile, miscible with water.
3. Steam volatile, miscible with water.
4. Not steam volatile, immiscible with water.
The aromatic structure(s) out of given structures is/are-
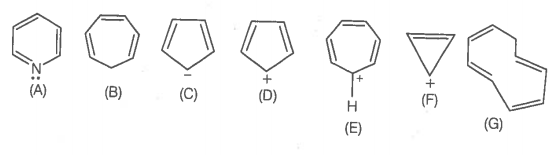
| 1. | A, C, D, F & G only | 2. | A & D only |
| 3. | A, C, E, F only | 4. | All are aromatic |
The Huckel's rule-based aromaticity is shown by:
| (A) |  |
(B) |  |
| (C) |  |
(D) |  |
| (E) |  |
(F) |  |
| 1. | A, B, D only | 2. | B, D only |
| 3. | B, D, E and F only | 4. | A, B, D, E & F only |
The type of structural isomerism shown by given compounds is-
and 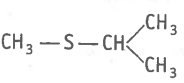
| 1. | Tautomerism | 2. | Positional isomerism |
| 3. | Functional isomerism | 4. | Ring Chain isomerism |
If a liquid compound decomposes at or below its boiling point, then the best method for purification is-
1. Distillation under reduced pressure
2. Azeotropic distillation
3. Gas chromatography
4. Sublimation
The most stable carbanion species among the following is-
| 1. | \(CCl^-_3\) | 2. | \(CH^-_3\) |
| 3. | \(CH_2Cl^-\) | 4. | \(CHCl^-_2\) |
The resonance hybrid structure will not exist for-
a. CH3OH
b. R - CONH2
c. CH3CH = CHCH2NH2
| 1. | a, and c | 2. | a, and b only |
| 3. | only a | 4. | b and c only |
Compare the stability of the two resonating structures given below and mark the correct option:
1. (I) is more stable than (II)
2. (II) is more stable than (I)
3. (I) and (II) both have the same stability
4. None of the above



