The hydrocarbons having the lowest dipole moment among the following is:
1. 
2. CH3 - C ≡ C - CH3
3. CH3 CH2 CH = CH2
4. CH2 = CH - C ≡ CH
1.

2. CH3 - C ≡ C - CH3
3. CH3 CH2 CH = CH2
4. CH2 = CH - C ≡ CH
The Cl — C — Cl angles in 1,1,2,2-tetrachloroethene and tetrachloromethane will be about :
1.
2.
3.
4.
The compound with an isopropyl group is-
1. 2,2,3,3-Tetramethylpentane
2. 2,2-Dimethylpentane
3. 2,2,3-Trimethylpentane
4. 2-Methylpentane
The enolic form of an acetone contains:
1. 9 sigma bonds, 1 pi bond, and 2 lone pairs of electrons
2. 8 sigma bonds, 2 pi bonds, and 2 lone pairs of electrons
3. 10 sigma bonds, 1 pi bond, and 1 lone pair of electrons
4. 9 sigma bonds, 2 pi bonds, and 1 lone pair of electrons
The compounds that show geometrical isomerism among the following are:
a. 2-Butene
b. Propene
c. 1-Phenylpropene
d. 2-Methylbut-2-ene
1. a, b
2. c, d
3. a, b, c
4. a, c
Arrange the following groups in order of decreasing - I (inductive) effect :
1.
2.
3.
4.
Incorrectly matched common name with IUPAC name is-
| Common name | IUPAC name |
| 1. Benzalacetophenone | (E)-1,3-Diphenylprop-2-en-1-one |
| 2. Glutaric acid | Pentenedioic acid |
| 3. Adipic acid | Hexane-1,6-dioic acid |
| 4. Pyruvic acid | 2-Oxopropanoic acid |
Which of the following organic compounds exhibit tautomerism?
| I. | 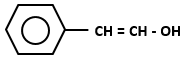 |
II. | 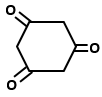 |
| III. | 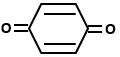 |
IV. | 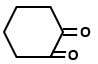 |
1. I and II
2. I, III and IV
3. I, II and IV
4. I, II, III and IV
The most stable carbocation among the following is-
| 1. | \(({CH_3})_3C\overset{+}{C}HCH_3\) | 2. | \({CH_3}CH_2\overset{+}{C}HCH_2CH_3\) |
| 3. | \(({CH_3})_2\overset{+}{C}CH_2CH_2CH_3\) | 4. | \({CH_3}CH_2\overset{+}{C}H_2\) |
The compound that is most difficult to protonate is:
| 1. |  |
2. |  |
| 3. |  |
4. |  |


