A compound that does not give a positive test in Lassaigne’s test for nitrogen is-
1.
Urea
2.
Hydrazine
3.
Azobenzene
4.
Phenyl hydrazine
The pair that represents chain isomers is-
| 1. | CH3CHCl2 and ClCH2CH2Cl | 2. | Propyl alcohol and Isopropyl alcohol |
| 3. | 2-Methylbutane and Neopentane | 4. | Diethyl ether and Dipropyl ether |

The IUPAC name of the above-mentioned compound is:
1. Pent-1-en-3-yne
2. Pent-1-ene-4-yne
3. Pent-4-yn-1-ene
4. Pent-1-en-4-yne
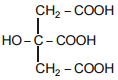
The IUPAC name of the above mentioned compound is -
1. Citric acid
2. 3-Hydroxy pentane-1,5-dioic acid
3. 2-Hydroxypropane-1,2,3-tricarboxylic acid
4. 2-Carboxy-2-hydroxy propane-1,3-dicarboxylic acid
The aromatic compound among the following is:-
| 1. | 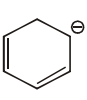 |
2. |  |
| 3. | 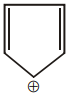 |
4. | 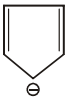 |
The pair of structures that does not represent isomers is:
| 1. | 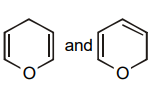 |
2. | 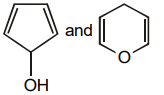 |
| 3. | 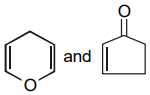 |
4. | 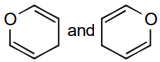 |
The effect that makes 2,3–dimethyl-2-butene more stable than 2-butene is-
1. Resonance
2. Hyperconjugation
3. Steric effect
4. Inductive effect
1. 2–Bromo-3–methylbutanoic acid
2. 2-Methyl-3-bromobutanoic acid
3. 3-Bromo-2-methylbutanoic acid
4. 3-Bromo-2,3-dimethylpropanoic acid.
The \(C - H\) bond distance is longer in -
| 1. | \(C_2H_2\) | 2. | \(C_2H_4\) |
| 3. | \(C_2H_6\) | 4. | \(C_2H_2Br_2\) |
The number of primary amines of the formula are:
1. 1
2. 3
3. 4
4. 5



