Select Chapter Topics:
The concentration vs time graph for the Haber process is given below:

The curve representing hydrogen gas is
1. Y
2. Z
3. X
4. In the Haber process, hydrogen gas is not used

Subtopic: Introduction To Equilibrium |
67%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Consider the following graph:
The point that represents re-established equilibrium is-
1. A
2. B
3. C
4. None of the above.
66%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | When Cu (II) and sulphide ions are mixed, they react together extremely quickly to give a solid. |
| Reason (R): | The equilibrium constant of Cu2+(aq) + S2- (aq) ⇋ CuS(s) is high because the solubility product is low. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A) |
| 2. | Both (A) and (R) are False. |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are True but (R) is not the correct explanation of (A) |
Subtopic: Solubility Product |
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
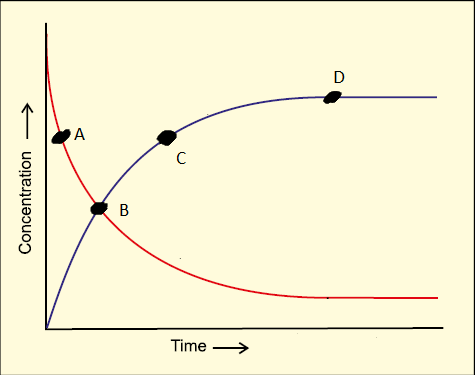
Consider the following graph:
The point that represents reaction in equilibrium is-
1. C
2. A
3. B
4. D
Subtopic: Introduction To Equilibrium |
51%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | For an exothermic reaction, the value of Kc decreases on increasing the temperature. |
| Reason (R): | Rate constant for a backward reaction increases more than the increase in the rate constant for a forward reaction. |
1. |
Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Subtopic: Kp, Kc & Factors Affecting them |
71%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Given below are two statements:
| Assertion (A): | In the dissociation of PCl5 at constant pressure and temperature, the addition of helium at equilibrium increases the dissociation of PCl5. |
| Reason (R): | Helium reacts with Cl2 . |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Kp, Kc & Factors Affecting them |
71%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | An aqueous solution of ammonium acetate can act as a buffer. |
| Reason (R): | Acetic acid is a weak acid and NH4OH is a weak base. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Buffer |
53%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Given below are two statements:
| Assertion (A): | An aqueous solution of ammonium carbonate is basic. |
| Reason (R): |
The acidic or basic nature of a salt solution of a salt of a weak acid and a weak base depends on the Ka and Kb values of the acid and the base forming it. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: pH calculation |
62%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | For any chemical reaction at a particular temperature, the equilibrium constant is fixed and is a characteristic property. |
| Reason (R): | Equilibrium constant is independent of temperature. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Kp, Kc & Factors Affecting them |
85%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Given below are two statements:
| Assertion (A): | The ionisation of hydrogen sulphide in water is low in the presence of hydrochloric acid. |
| Reason (R): | Hydrogen sulphide is a weak acid. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Common Ion Effect |
53%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch