For the reaction
\(C H_{3} C O O H \left(\right. l \left.\right)+ 2 O_{2} \left(\right. g \left.\right) \)
\(\rightleftharpoons 2 C O_{2} \left(\right. g \left.\right)+ 2 H_{2} O \left(\right. l \left.\right)\)
at 25°C and 1 atm. pressure, ΔH = –874 kJ. The change in internal energy (ΔE) is:
1. – 874 kJ
2. – 971.53 kJ
3. + 971.53 kJ
4. + 874 kJ
\(\rightleftharpoons 2 C O_{2} \left(\right. g \left.\right)+ 2 H_{2} O \left(\right. l \left.\right)\)
at 25°C and 1 atm. pressure, ΔH = –874 kJ. The change in internal energy (ΔE) is:
1. – 874 kJ
2. – 971.53 kJ
3. + 971.53 kJ
4. + 874 kJ
The occurrence of a reaction is impossible if:
1. ΔH is +ve ; ΔS is also + ve but ΔH < TΔS
2. ΔH is – ve ; ΔS is also – ve but ΔH > TΔS
3. ΔH is – ve ; ΔS is + ve
4. ΔH is + ve ; ΔS is – ve
The unit of entropy is -
1. J mol–1
2. JK mol–1
3. J mol–1 K–1
4. J–1 K–1 mol–1
Combustion of glucose takes place according to the equation,
,
Energy required for the production of 1.6 g of glucose is -
(Molecular mass of glucose = 180 g)
1. 0.064 kcal
2. 0.64 kcal
3. 6.4 kcal
4. 64 kcal
When a gas undergoes adiabatic expansion, it gets cooled due to -
1. Loss of energy
2. Fall in pressure
3. Decrease in velocity
4. Increase in energy with work done
A Beckmann thermometer is used to measure -
1. High temperature
2. Low temperature
3. Normal temperature
4. All temperatures
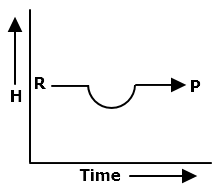
Which plot represents an exothermic reaction?
1.
2.
3.
4.
The work done in ergs for the reversible expansion of one mole of an ideal gas from a volume of 10 liters to 20 liters at 25°C is -
1.
2.
3.
4.
Which of the following is not a state function?
| 1. | ΔS | 2. | ΔG |
| 3. | ΔH | 4. | ΔQ |