For the graph given below, it can be concluded that work done during the process shown will be-

1.
Zero
2.
Negative
3.
Positive
4.
Cannot be determined

Consider the following graph.

The work done, as per the graph above, is:
| 1. | Positive | 2. | Negative |
| 3. | Zero | 4. | Cannot be determined |
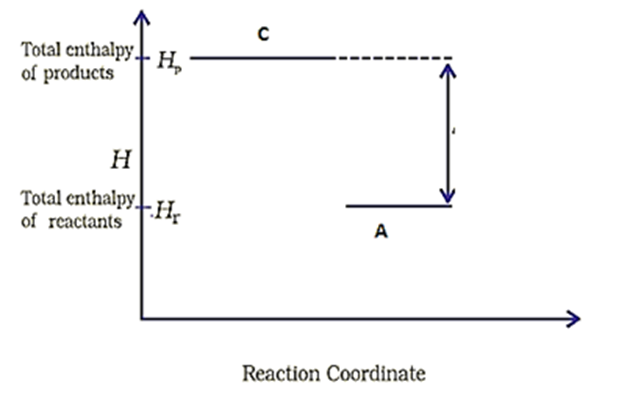
Consider the following diagram for a reaction .
The nature of the reaction is-
1. Exothermic
2. Endothermic
3. Reaction at equilibrium
4. None of the above
What is the nature of the reaction depicted in the given diagram for A→C?

| 1. | Exothermic | 2. | Endothermic |
| 3. | Reaction at equilibrium | 4. | None of these |
An enthalpy diagram for a particular reaction is given below:
Which of the following statements is correct?
| 1. | Reaction is spontaneous |
| 2. | Reaction is non-spontaneous |
| 3. | The spontaneity of the reaction cannot be determined from the graph provided above. |
| 4. | None of the above |
The entropy change in the surroundings when 1.00 mol of H2O(l) is formed under standard conditions is:
∆fHθ = –286 kJ mol–1
1. 952.5 J mol-1
2.
3.
4.
The thermodynamic stability of NO(g) based on the above data is:
1. Less than NO2(g)
2. More than NO2(g)
3. Equal to NO2(g)
4. Insufficient data
The difference between enthalpy (∆H) and internal energy (∆E) for the given below reaction,
under constant temperature, is:
\(C_{3} H_{8} \left(\right. g \left.\right) + 5 O_{2} \left(\right. g \left.\right) \rightarrow 3 CO_{2} \left(\right. g \left.\right) + 4 H_{2} O \left(\right. l \left.\right)\)
| 1. | + RT | 2. | – 3RT |
| 3. | + 3RT | 4. | – RT |
For the given reaction
, the heat of formations of are -188 kJ/mol & -286 KJ/mol respectively. The change in the enthalpy of the reaction will be:
1. – 196 kJ/mol
2. + 196 kJ/mol
3. + 948 kJ/mol
4. – 948 kJ/mol
| Assertion (A): | Work done in an irreversible isothermal process at constant volume is zero. |
| Reason (R): | Work is assigned a negative sign during expansion and is assigned a positive sign during compression. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | Both (A) and (R) are false. |