A sample containing 1.0 mol of an ideal gas is expanded isothermally and reversible to ten times of its original volume in two separate experiments. The expansion is carried out at 300 K and at 600 K respectively.
The correct statements among the following are:
(a)
Work done at 600 K is 20 times the work done at 300 K.
(b)
Work done at 300 K is twice the work done at 600 K.
(c)
Work done at 600 K is twice the work done at 300 K.
(d)
∆ U = 0 in both cases.
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, d)
The correct statements among the following are :
| a. | The enthalpy of two moles of ZnO is less than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ |
| b. | The enthalpy of two moles of ZnO is more than the total enthalpy of two moles of Zn and one mole of oxygen by 693.8 kJ |
| c. | 693 . 8 kJ mol -1 energy is evolved in the reaction |
| d. | 693 . 8 kJ mol -1 energy is absorbed in the reaction |
1. (a, b)
2. (b, c)
3. (c, d)
4. (a, c)
For the graph given below, it can be concluded that work done during the process shown will be-
| 1. | Zero | 2. | Negative |
| 3. | Positive | 4. | Cannot be determined |
Consider the following graph.
The work done shown by the above-mentioned graph is-
| 1. | Positive | 2. | Negative |
| 3. | Zero | 4. | Cannot be determined |
Consider the following diagram for a reaction .
The nature of the reaction is-
1. Exothermic
2. Endothermic
3. Reaction at equilibrium
4. None of the above
Consider the following diagram for a reaction
The nature of the reaction is-
| 1. | Exothermic | 2. | Endothermic |
| 3. | Reaction at equilibrium | 4. | None of the above |
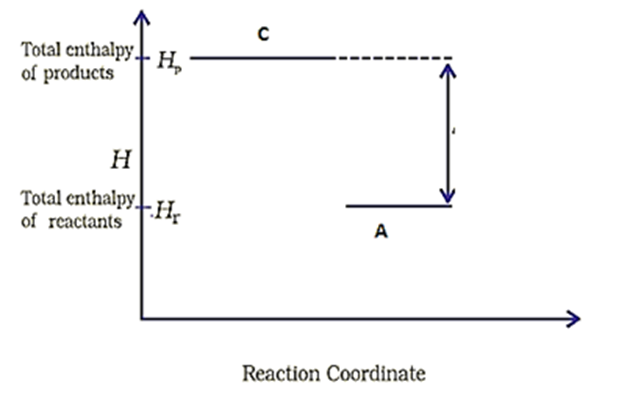
An enthalpy diagram for a particular reaction is given below:
The correct statement among the following is-
1. Reaction is spontaneous
2. Reaction is non-spontaneous
3. Cannot predict spontaneity of the reaction from the graph given above
4. None of the above
1.0 mol of a monoatomic ideal gas is expanded from state (1) to state (2) as shown in the graph below:
The work done for the expansion of gas from state (1) to state (2) at 298 K will be
1. 1617.6 J
2. -1617.6 J
3. 1717.6 J
4. -1717.6 J
| Column I | Column II | ||
| a. | No heat is absorbed by the system from the surroundings, but work (w) is done on the system. | i. | ∆U = q – w, closed system. |
| b. | No work is done on the system, but q amount of heat is taken out from the system and given to the surroundings. | ii. | ∆U = wad, a wall is adiabatic. |
| c. | w amount of work is done by the system and q amount of heat is supplied to the system. | iii. | ∆U = –q, thermally conducting walls. |
| 1. | a = i; b = ii; c = iii | 2. | a = ii; b = i; c = iii |
| 3. | a = ii; b = iii; c = i | 4. | a = iii; b = ii; c = i |
| (i) | When liquid crystallizes into a solid, entropy increases. |
| (ii) | When the temperature of a crystalline solid is raised from 0 K to 115 K then entropy increases. |
| (iii) | 2 NaHCO3 (s) →Na2CO3 (s) +CO2(g)+H2O(g); Entropy increases. |
| (iv) | H2(g)→2H(g) ; Entropy decreases. |