An electronic configuration representing an atom in the excited state is:
Consider the following sets of quantum numbers:
n l m s
(i) 3 0 0 +1/2
(ii) 2 2 1 +1/2
(iii) 4 3 -2 -1/2
(iv) 1 0 -1 -1/2
(v) 3 2 3 +1/2
Which of the following sets of quantum numbers is not possible?
1. ii, iii and iv
2. i, ii, iii and iv
3. ii, iv and v
4. i and iii
If uncertainty in position and momentum are both equal, then uncertainty in velocity will be:
1.
2.
3.
4.
The group number and electronic configuration of element Z =114 is:
1. Halogen family, [Rn]5f146d107s27p5
2. Carbon family, [Rn]5f146d107s27p2
3. Oxygen family, [Rn]5f146d107s27p4
4. Nitrogen family, [Rn]5f146d107s27p6
When an electron jumps from n=5 to n=1 in a hydrogen atom, the number of spectral lines obtained is
| 1. | 3 | 2. | 4 |
| 3. | 6 | 4. | 10 |
Incorrect statement among the following is:
| 1. | The uncertainty principle is\(\Delta x \cdot \Delta p \geq \dfrac{h}{4 \pi} \) |
| 2. | Half-filled and fully filled orbitals have greater stability due to greater exchange energy, greater symmetry, and a more balanced arrangement. |
| 3. | The energy of the 2s orbital is less than the energy of the 2p orbital in the case of hydrogen-like atoms. |
| 4. | De-Broglie's wavelength is given by\(\lambda=\dfrac{h}{m v} \), where m= mass of the particle, v = group velocity of the particle. |
The incorrect statement among the following is:
| 1. | Total orbital angular momentum of an electron in 's' orbital is equal to zero. |
| 2. | An orbital is designated by three quantum numbers, while an electron in an atom is designated by four quantum numbers. |
| 3. | The electronic configuration of N atom is 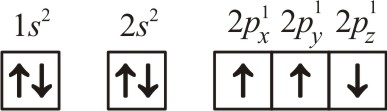 |
| 4. | The value of m for dz2 is zero. |
The correct set of four quantum numbers for the valence electron of a rubidium atom (Z =37) is:
| 1. | \(5,1,1,+ {1 \over2}\) | 2. | \(6,0,0,+ {1 \over2}\) |
| 3. | \(5,0,0,+ {1 \over2}\) | 4. | \(5,1,0,+ {1 \over2}\) |
The incorrect set of quantum numbers among the following is:
1. n= 4, l= 0, m= 0, s= -1/2
2. n= 5, l= 3, m= 0, s= +1/2
3. n= 3, l= 2, m= -3, s= -1/2
4. n= 3, l= 2, m= 2, s= -1/2
The correct order of the total number of nodes of atomic orbitals is:
1. 4f > 6s > 5d
2. 6s > 5d > 4f
3. 4f > 5d > 6s
4. 5d > 4f > 6s


