Select Chapter Topics:
Assertion (A):
In the third energy level, there is no f subshell.
Reason (R):
For n = 3, the possible values of l are 0, 1, 2 and for f subshell l = 3.
1.
Both (A) and (R) are true and (R) is the correct explanation of (A).
2.
Both (A) and (R) are true but (R) is not the correct explanation of (A).
3.
(A) is true but (R) is false.
4.
(A) is false but (R) is true.
Subtopic: Shell & Subshell |
78%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | The angular momentum of an electron in an atom is quantized. |
| Reason (R): | In an atom, only those orbits are permitted in which the angular momentum of the electron is a natural number multiple of h/2π. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Bohr's Theory |
80%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | The radius of the second orbit of He+ is equal to that of the first orbit of hydrogen. |
| Reason (R): | The radius of an orbit in hydrogen-like species is directly proportional to n and inversely proportional to Z. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of the (A). |
| 2. | Both (A) and (R) are True and (R) is not the correct explanation of the (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Subtopic: Bohr's Theory |
71%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | The number of radial nodes in 3 dz2 , is zero. |
| Reason (R): | The number of angular nodes of atomic orbitals is equal to the value of l. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | (A) is False but (R) is True. |
Subtopic: Nodal Plane |
67%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
| Assertion (A): | Bohr’s orbits are also called stationary states. |
| Reason (R): | Electrons are stationary in an orbit. |
| 1. | Both (A) and (R) are true and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are true but (R) is not the correct explanation of (A). |
| 3. | (A) is true but (R) is false. |
| 4. | (A) is false but (R) is true. |
Subtopic: Bohr's Theory |
69%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
Links
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
The correct statement(s) among the following regarding quantum numbers is -
| a. | Angular quantum number determines the three-dimensional shape of the orbital |
| b. | The principal quantum number determines the orientation and energy of the orbital |
| c. | Magnetic quantum number determines the size of the orbital |
| d. | Spin quantum number of an electron determines the orientation of the spin of the electron relative to the chosen axis |
Choose the correct option:
1. (a), (d)
2. (b), (c)
3. (c), (d)
4. (b), (d)
Subtopic: Quantum Numbers & Schrodinger Wave Equation |
72%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Consider the following graph.
The correct relation between T1 and T2 is-
1. T1 > T2
2. T1 < T2
3. T1 = T2
4. None of the above
Subtopic: Photo Electric Effect |
78%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch

The ultraviolet region in the above figure is indicated by:
1. D
2. B
3. A
4. C
Subtopic: Introduction of Atomic Structure |
73%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
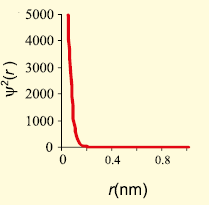
The graph that represents the probability density vs distance of the electron from the nucleus for 2s orbital is -
1.
2.
3.
4.
Subtopic: Quantum Numbers & Schrodinger Wave Equation |
67%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
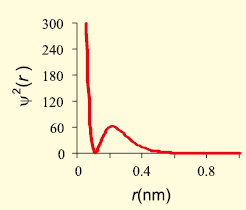
The graph that represents the variation of (probability of density of finding the electron) with distance (r) is given below:
The number of nodes as per the graph is-
1. zero
2. 1
3. 2
4. Cannot predict
Subtopic: Shell & Subshell |
63%
From NCERT
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Hints
To view explanation, please take trial in the course.
NEET 2025 - Target Batch
Select Chapter Topics: