The relation between two specific heats (in cal/mol) of a gas is:
1.
2.
3.
4.
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe ratio of the specific heats in terms of degrees of freedom (\(n\)) is given by:
1. \(1+1/n\)
2. \(1+n/3\)
3. \(1+2/n\)
4. \(1+n/2\)
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology| Assertion (A): | The ratio \(C_p\over C_v\) is more for helium gas than for hydrogen gas. |
| Reason (R): | Atomic mass of helium is more than that of hydrogen. |
| 1. | Both (A) and (R) are True and (R) is the correct explanation of (A). |
| 2. | Both (A) and (R) are True but (R) is not the correct explanation of (A). |
| 3. | (A) is True but (R) is False. |
| 4. | Both (A) and (R) are False. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
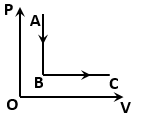
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe figure shows a process for a gas in which pressure (P) and volume (V) of the gas change. If and are the molar heat capacities of the gas during the processes AB and BC respectively, then:
1.
2.
3.
4.
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe specific heat of an ideal gas is:
1. proportional to
2. proportional to T2.
3. proportional to T3.
4. independent of
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyIf \(C_p\) and \(C_v\) denote the specific heats (per unit mass) of an ideal gas of molecular weight \(M\) (where \(R\) is the molar gas constant), the correct relation is:
1. \(C_p-C_v=R\)
2. \(C_p-C_v=\frac{R}{M}\)
3. \(C_p-C_v=MR\)
4. \(C_p-C_v=\frac{R}{M^2}\)
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyFor hydrogen gas \(C_P-C_V=a\) and for oxygen gas \(C_P-C_V=b\) where molar specific heats are given. So the relation between \(a\) and \(b\) is given by: (where \(C_p\) and \(C_V\) in J mol-1 K-1)
1. \(a=16b\)
2. \(b=16a\)
3. \(a=4b\)
4. \(a=b\)
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe specific heat of a gas:
| 1. | has only two values \(Cp\) and \(Cv\). |
| 2. | has a unique value at a given temperature. |
| 3. | can have any value between 0 and ∞. |
| 4. | depends upon the mass of the gas. |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe value of for a gas in state A and in another state B. If denote the pressure and denote the temperatures in the two states, then:
| 1. | \(P_A=P_B ; T_A>T_B\) |
| 2. | \(P_A>P_B ; T_A=T_B\) |
| 3. | \(P_A<P_B ; T_A>T_B\) |
| 4. | \(P_A=P_B ; T_A<T_B\) |
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & BiologyThe amount of heat energy required to raise the temperature of \(1\) g of Helium at NTP, from \({T_1}\) K to \({T_2}\) K is:
1. \(\frac{3}{2}N_ak_B(T_2-T_1)\)
2. \(\frac{3}{4}N_ak_B(T_2-T_1)\)
3. \(\frac{3}{4}N_ak_B\frac{T_2}{T_1}\)
4. \(\frac{3}{8}N_ak_B(T_2-T_1)\)
Prefer Books for Question Practice? Get NEETprep's Unique MCQ Books with Online Audio/Video/Text Solutions via Telegram Bot
NEET MCQ Books for XIth & XIIth Physics, Chemistry & Biology